JOURNAL OF COSMETIC SCIENCE 120 DELIPIDATION OF HAIR Using a Soxhlet extraction apparatus, free internal and surface lipids were removed from hair. This method is based on an established procedure in which hair is extracted with a series of solvents of increasing polarity (41). The apparatus consisted of a round-bottom fl ask to which a Soxhlet extraction tube was mounted. Inside the Soxhlet extraction tube, a bundle of hair was placed in a cellulose thimble. A condenser was mounted on top of the Soxhlet extraction tube. The effect of solvent extraction on hair was investigated fi rst by treatment with t-butanol and n-hexane for 4 h each, then with a mixture of chloroform/ methanol (70:30, v/v) for 6 h. In each procedure, 3 g of hair was treated with 250 mL of solvent in the Soxhlet extractor. PREPARATION OF HAIR CROSS SECTIONS Hair fi bers were embedded in an epoxy resin block. The hair-epoxy block was cut into approximately 2-mm-thick sections. A Buehler MetaServ 250 Grinder-Polisher (Lake Bluff, IL) was used to prepare a smooth surface of the hair cross sections, suitable for AFM analysis. In this procedure, the hair-epoxy sections were sequentially grinded with alumi- num oxide sandpaper with grit levels of 300, 600, and 1,200 with a head force set to 0.5 lb (|2.22411 N). Both the head and plate rotate at a speed of 150 rpm in opposite directions. The lower head force and spin rate ensure mild abrasion and no tearing of the hair specimen. After sandpaper treatment, polish was applied with a liquid suspension of Al2O3 (0.05 μm). Suspended Al2O3 particles can roll and gently slide across the cloth and specimen. Finally, the hair-epoxy section was sonicated in deionized water for 1 min to eliminate the residues generated from grinding and polishing. This polishing technique was chosen as an alternative to conventional microtome preparation, which can introduce a variety of artifacts, including scores and tearing, chatters (thick and thin zones parallel to the knife edge), and compression artifacts. If performed incorrectly (e.g., with a large head force or high spinning speed), grinding and polishing can also introduce artifacts to the sample resulting in scratches, tearing, and uneven surfaces. Polishing is a preferential technique for preparing various biological specimens, such as bone or tooth, for micro- scopic investigations. Because a rough surface prevents the AFM probe from being able to raster effectively over the sample, the polishing technique creates a smooth surface and makes the sample more amenable for AFM analysis. AF M MEASUREMENTS Co ntact-mode AFM studies were carried out using a Multimode Nanoscope III (Bruker Corporation, Billerica, MA) using a 128-μm piezoelectric scanner. In this technique, a small tip attached to a cantilever is rastered in the X and Y directions on the surface of a sample, and the height (Z direction) is monitored. The operation of the instrument is based on the principal that a diode laser beam is focused on the top of a cantilever and refl ected onto a position-sensitive photodetector. The probe (cantilever and tip) is shaped like a stylus, and as it traverses across the surface, the topographic features are mapped based on forces encountered between the probe and the sample. An Si3N4 probe (SNL-10) with a triangular cantilever (force constant of 0.06 N/m) from Bruker Corporation was used for all experiments. We operated the instrument in constant force mode, in which the
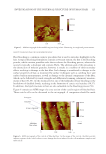
INVESTIGATION OF THE INTERNAL STRUCTURE OF HUMAN HAIR 121 sample vertical height is continuously adjusted via an electronic feedback loop so that the probe maintains a constant force on the sample. All of the AFM micrographs presented in this article are raw images, which have not been fi ltered in any way, and are representa- tive of many measurements carried out on each data set. AFM (and other microscopies) can sometimes be misleading because of the small region represented in an image. For this reason, we scout many areas to ensure that the selected images are representative of the data set. In the fi nal analysis, at least three hair fi ber cross sections were analyzed for each hair treatment set, and images were collected in a minimum of four sections of each cross section. It should be noted that care must be taken to ensure that enough fi bers are sampled, and suffi cient area of each fi ber is analyzed to provide an accurate depiction of the morphological condition of the fi ber. Each AFM image represents a small area of the hair fi ber, and one can fi nd a pristine or extremely damaged region on the same fi ber. RES ULTS AND DISCUSSION The evaluation of cross sections of hair is a novel tec hnique that provides insight into the morphological properties of hair and the effects of damaging treatments and environmental exposure. We demonstrate the use of AFM analysis of hair cross sections to identify key structural components of the cuticle and cortex of hair while also offering insight into its structural architecture, in agreement with other recently published results. We also provide key insights into induced changes to the internal structural components of hair by hair bleaching, one of the most common chemical processes used in hair salons. In addition, we examine the effects of removing endogenous and exogenous lipids from hair and identify unique details that provide insight into the boundary region surrounding macrofi brils. IDE NTIFICATION OF ULTRAFINE STRUCTURAL FEATURES IN HAIR CROSS SECTIONS Fig ure 1 contains an optical micrograph with an inset that roughly approximates the region of a hair cross section where the AFM micrograph was collected. Immediately apparent in Figu re 1. Typical AFM micrograph of a cross section of human hair. Note that only a section of the hair cross section is visible in the fi eld of view of the image. The bright-fi eld microscope image (refl ection mode) of several cross sections of virgin hair is provided to give a scale of the section imaged by AFM.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)