306 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS x 1041 10 Of , • Squalane 1 O0 br-C8 -Trigly 0 5O 0 50 100 Component of Oil phase % (w/w) Figure 19. Influence of a component in oil phase on stability ofo/w emulsions
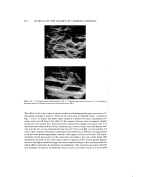
WATER-IN-OIL EMULSIONS 307 Figure 20. Model of emulsion obtained by gel-emulsification method The fact that in the case of acidic amino acids and basic amino acids, the best gels were obtained only when the monosodium salts or monohydrochlorides, respectively, was considered to be closely related to the fact that the molecules were in the highest possi- ble polyvalent state (lid among the 4 in the aqueous solution as is shown below. (I) (II) (III) (IV) CO2H CO2H COO- COO- H•N+--GIu--CO/H • H•N--Glu--COO • H•N--GIu--COO-•- H/N--GIu--COO- pK 2.2 3,2 4.3 9,7 In the case of neutral amino acids, the i'soelectric range was also found to be the most suitable for the formation of the gels. When one note the relationship between the solubility (6) and lOB as is shown in Fig. 4, it becomes apparent that the higher the solubilities in water the better the results ob- tained. The lOB is within the range of about 4 to 10. With acylated amino acids and their higher alcohol esters, there were no gel formations. Various gels were obtained by a variety of combinations between amino acids and' surfactants, such as their kinds and mixing ratio. The higher the concentration of amino acid, and it's mixing ratio to the surfactant and shearing stress the more stable was the gel obtained. In this case, the hardness and viscoelasticity of the gels increased and the gels became transparent. On the contrary, when the concentration and mixing ratio be- came lower, the gel appeared milky white and fluid. Figure 5 shows the theological properties of the gels obtained between Sunsoft O-30B and a 10 per cent aqueous so-
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)