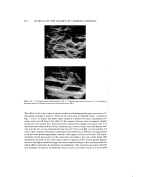
304 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Table V The Properties of the Creams Obtained from the Gels Prepared by the Addition of an Amino acid, and without an Amino acid Physical Properties of Cream Stability of Cream Viscosity Hardness Emulsion After 1 Day Number 70øC 30øC 0øC 25øC 3]øC Particles 0øC 25øC 37øC After 30 Days 0øC 25øC 37øC cps (1) a 18000 94400 42 11 6 (See Fig. 18(a)) o o o (2) b 13900 101600 53 15 7 o o o (3) e 8500 36000 12 3 1 (See Fig. 18(b)) (4) • 5750 38400 15 4 1 O' o o o o o o , x • X x : Evaluation:good*--o g} a x X•poor. a(1) 40 per cent aqueous solution of monosodium L-glutamate monohydrate•Sunsoft © O-30B. b(2) 40 per cent aqueous solution of monosodium L-glutamare monohydrate-POEM © O-72-D. c(3) Water- Sunsoft © O-30B. •(4) Water-POEM © O-72-D. Table VI The Relationship of the X-Ray Diffraction Patterns of the Surfactanrs to their Gel-Forming Function and the Properties of W/O Emulsions using the Gels Surfactant X-Ray W/O Emulsion's Diffraction Gel- Stability Viscosity Name Pattern Formation (RT) (30øC) Sunsoft © O-30B a Clear o o 94400 H DIG-EIS b Clear o o 112800 Emalex © EG-O c Indistinct x X -- (two-phase separations) i•:il ' Eraalex © 300dxo ½ Indistinct x x 1750 Emalex © 503 c No'•peak x x 750 Nikkol © MIO-2 '• No peak x X ::':":• (two-phase separations) '•. •Taiyo Kagaku Co., Ltd. (62 Akahori, Yokkaichi, Mie, Japan). bMatsumoto Trading Co., Ltd. (3-1 Nihonbashihoncho, Chuoku, Tokyo, Japan). •5 Nihon Emulsion Co., Ltd. (5-32-7 Minami, Koenji, Suginamiku, Tokyo,Japan). aNikko Chemicals Co., Ltd. (1-4-8 Bakurocho, Nihonbashi, Chuoku, Tokyo, Japan).
WATER-IN-OIL EMULSIONS 305 ß _. ' (a) , (b) Figure 18. Comparison of emulsion particles of creams: division, 2.5rtm acid, linolic acid, and isohexadecanoic acid. These suroeactants are supplied in the form of a mixture of partial esters. For example, when those with 3 hydroxyl groups were used, a mixture comprised mainly of monoester was preferred, and when those with 4 hydroxyl groups were used, a mixture comprising ofdiester gave the better results. On the other hand, even within the above mentioned range, those in which the hydroxyl groups were completely esterified, failed to form gels. Although, those surfactants which formed gels did not contain water, all of them gave 2 clearly discernible diffrac- tion lines in the small angle region as seen in Fig. 3. As is seen in Table I, the spacings of the surfactants appeared at approximately 33 and 70 •, and the ratio was about 1:2. From these facts, it was considered that, although, they were liquids, these surfactants possessed very orderly lameliar structures by themselves. On the other hand, almost all of those surfactants lacking the function of gel formation did not have a clear structure, as is shown in Table II. The X-ray diffraction patterns were indistinct or completely lacking even though they were clear the spacings were either too wide or too narrow. Such results of X-ray analysis were identified as having a close correlation to gel forma- tion. As a result, it can be concluded that, in. order to form gels, the corresponding surfactants are required to have at least a high orderly lameliar structure. Contrary to the restriction of the surfactants used, almost all of the amino acids and their salts formed gels. As shown in Table III, nearly all of the monoamino- rnonocarboxylic acids (neutral amino acid) with an. isoelectric point in the. weak acidic range, such as monosodium salts of monoamino-dicarboxylic acid (acidic amino acid) and monohydrochlorides of diamino-monocarboxylic acid (basic amino acid) were ca- pable of forming gels. Those readily soluble in. water were very effective, such as glycine, L-alanine, B-alanine, hydroxy L-proline, L-serine, monosodium Dglutamate rnonohydrate, monopotassium L-glutamate monohydrate, monosodium L-aspartate monohydrate, monopotassium Daspartate dihydrate, L-lysine monohydrochlo/-ide L-arginine monohydrochloride, and L-histidine monohydrochloride. The D- and DL stereoisomers also gave good gel formations.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)