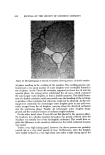
EMULSION STABILITY 811 Figure 14. Microphotographs of oleyl alcohol emulsions Top. Surfactant initially in oil Left to right. Emulsions O-1, 0-2, 0-3 Bottom. Suffactant initially in water Left to right. Emulsions W-l, W-2, W-3 at various stages of emulsification. Some of the examples are shown in Fig. 14. Examination of these microphotographs indicates the presence of a large number of double emulsions of (W/O)/W type in the emulsions prepared by initially placing the surfactant in the oil phase (i.e., Emulsion O series). As shown in the photograph O-1, which was taken at an early stage of emulsi- fication, extremely small water droplets were observed in very large oil drops. Sometimes the boundaries of these oil drops were not well-defined at this stage. The picture O-2 clearly indicates the presence of double emulsion droplets. These double emulsion droplets disappeared after a prolonged mix- ing as shown in 0-3. Double emulsions were also observed in some emulsions prepared by initially placing the surfactant in the aqueous phase (i.e., Emul- sion W series) but the number of droplets containing the double emulsion was much lower. A close examination of the microphotographs suggests that the formation of the double emulsion might be the reason for the formation of fine emulsion even under very slow mixing when the surfactant was placed in the oil phase. An enlarged photograph of such a double emulsion is shown in Fig. 15. One possible mechanism is that when the emulsion was made by first placing Tween 80 in the oil phase, water initially entered the surfactant micelies in the
812 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Figure 15. Microphotograph of mineral oil emulsion showing presence of double emulsion oil phase resulting in the swelling of the micelies. The swelling process con- tinued and a very great number of water droplets were eventually formed in the oil phase. As the Tween 80 molecules migrated out from the oil into the aqueous phase, the mixing action subdivided the oil mass, which contained the microscopic •vater droplets, to form a double emulsion. The initial forma- tion of a double emulsion could conceivably ease the breaking of the oil drops to produce a finer emulsion that otherwise could not be obtained. As the mix- ing process continued, the microscopic water droplets grew in size and even- tually escaped from the oil droplets, carrying along the dissolved surfactant into the conth•uous phase. Finally, all microscopic water droplets disap- peared and the emulsion became an ordinary single emulsion. On the other hand, when T•veen 80 was first placed in the aqueous phase, the tendency for a double emulsion formation was greatly reduced since the oil phase was initially free of the hydrophilic surfactant. This would then ex- plah• the difference in the emulsions obtained as the initial surfactant location was varied. Under a very high mixing speed, ho•vever, the above process was probably carried out in a very short period of ti•ne. Furthermore, since the droplets were readily broken by a very high shear rate under a high mixing speed, the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)