WATER-IN-OIL EMULSIONS 309 obtained by DTA and optical microscopic observations. From Fig. 6, it can be said that the gel itself is considered to be the same emulsion system as an ordinary w/o emulsion in which the particles of aqueous solution are dispersed in the lipophilic surfactant phase. It can be readily understood, as is shown in Fig. 5, that fluidity, high viscosity, and hardness of the gel are due to the increasing volume ratio and interfacial area. In the gels obtained by the addition of aqueous solution of amino acids, the interfacial films are very strong and stable so that the particles do not coalesce even though the in- ner phase is increased. These phenomena owe much to the functions of the amino acid. From these facts, the authors studied the influence of the amino acids on the change of the lameliar structure possessed in common with the surfactants used. Figure 9 indicates the comparison between the spacings of stable gels with aqueous so- lutions of amino acid and those of unstable gels with water only and urea. Sunsoft O-30B, which was used as the surfactant in the experiments, had spacings of 33.8 ,•. In the case of an aqueous solution of amino acid, even though the mixing ratio of the gel was altered, the original spacing of the surfactant did not change. However, in the case of water, it increased to 37.6 •. Such change had been determined in either case at an early stage, regardless of the increased mixing ratio. It may be considered that the " amino acid results in weakening the interaction between the water and the surfactant : that is, the amino acid decreases the solubility of the surfactant in the water phase and also prevents the solubilization of water into the hydrophilic parts of the lameliar struc- ture, therefore, it seems to prevent the change in the original structure of the ::"" surfactant. It was found that urea further increased the spacing than in the case of :. water. Urea formed a gel with difficulty and though formed, was immediately destroyed and inverted to the o/w type. The explanation of the results of NMR measurement are shown in Fig. 10 and shows how the amino acids or their salts interact with water. A negative sign Ad indicates a • shift of proton in water to the lower magnetic field, and a positive sign means a shift to the higher magnetic field. The amino acids allows the proton in water to shift to the ß :: lower magnetic field. The gradient was sharp, hence, the effect was much greater even in lower concentration. On the contrary, potassium thiocyanate (KSCN) and urea :- •: shifted the proton to the higher magnetic field. In other words, these facts suggest a change in the physical properties of water. An amino acid, which shifts the proton to the lower magnetic field, indicates a condition in which the affinity of water to the amino acid is very strong and results in a decrease of free water. Therefore, these •:i amino acids decreased the solubility of surfactant, which previously existed in water or prevented the solubilization of water in the hydrophilic part of the surfactant. These concepts are supported by the data obtained in the heat of solution shown in Fig. 8. Furthermore, since the amino acid will expel the surfactant from the water phase, it be- comes evident that a w/o type emulsion is the easier one to obtain. It is presumed that there is a decrease in the PIT with the increase concentration of the amino acids. This hypothesis was confirmed by the results of the PIT as is shown in Fig. 11. The amino acids decreased the PIT, and conversely, KSCN and urea increased the PIT. Such a trend in the PIT corresponded perfectly with the results of the NMR analysis. From this view point, it can be readily understood how the amino acid changes the physical properties of water and decreases the interaction with the surfactant. In order to inves- tigate how much water causes a change of structure when solubilized in the hydrophilic part of the surfactant, the authors compared the changes in the amount of water migrat-
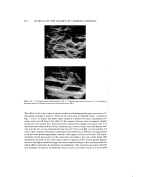
310 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS ing into the surfactant phase with that of the spacings. Figs. 12 and 13 show the changes in the water content caused by its migration into the surfactant phase with a lapse in time. In the case of water only, the migration to the surfactant phase was as much as 6 per cent. In case of monosodium L-glutamate monohydrate, the amount of migration decreased with an increase in concentration. This corresponds well with the decreasing trend of affinity between the surfactant and water. The water content became constant rapidly and did not increase over a prolonged period. The relationship of this water content to the spacings and the stability of the gels is in- dicated in Table IV. As is evident from this Table, with the increase of water migrating to the surfactant, the spacing becomes much wider. The spacing obtained with 40 per cent aqueous solution of monosodium L-glutamate monohydrate was exactly the same as that of the surfactant itself (Sunsoft O-30B). No changes in the structure of the surfactant was observed when the solubilized water was less than 3 per cent. The larger the variation of the spacings, the poorer the gel formation became. The effect of pH on the stability of the gel can also be explained by the variation of water content and the spacings as shown in Figs. 14 and 15. As the pH of water increased, the water solubilized in the surfactant increased, and at the same time the spacings also increased rapidly. From the above results and discussions, it is possible to draw the structural model indi- cating the mechanism for the stabilization of the gels by the amino acids as is shown in Fig. 16. The upper models indicate the overall view, and the lower models show a magnified view. The surfactant, though it is in the liquid state, has a lameliar structure as seen in view (1) of Fig. 16. In the case of the aqueous solution in the absence of amino acids, a large amount of water is solubilized in the hydrophilic part of the surfactant having an orderly form and its original structure becomes disordered and loosened due to the widening of its spacings. Under such conditions, water particles coalesce easily, which indicates instability. The same explanation can be made in the case of KSCN and urea, which change the structure of the surfactant. This is shown in view (2) of Fig. 16. View (3) of Fig. 16 indicates the case when amino acid is added. Since the amount of water to be solubilized in the hydrophilic part of the surfactant is limited and does not induce any change of structure, a concentrated and tight inter- facial atmosphere is established around the particles. As a result, coalescence of the particles hardly occurs thus, maintaining stability. As an important fact to support such a structural model, we succeeded in taking the photograph of the gel by EM (as is seen in Fig. 17). The sample shown was the gel obtained with Sunsoft O-30B and a 40 per cent aqueous solution of monosodium L-glutamate monohydrate mixed in the ratio of 1:4. Apparently, the water particles surrounded by the surfactant phase in the lamellar structure can be seen. GEL-EMULSIFICATION METHOD The relationship between the stability of the gels and that of w/o emulsions made by using the gels has also been noted. Hardness of the gels was influenced by the shear stress given at the time of preparation. When the hardness of the gels was changed, the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)