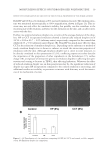
MOISTURIZING EFFECTS OF HYDROGENATED POLYISOBUTENE 23 (TEWL) or (b) by increased occlusion, thereby causing a decrease in TEWL. When we measured TEWL upon application of HP, as well as of CCT, we observed that the TEWL decreased signifi cantly, proving the effectiveness of HP and CTT in retaining skin hydra- tion. This effect was longer-lasting in the case of HP and was maintained at a 30% lower level as compared to untreated skin for up to 6 h after application. Such reduction in TEWL has been reported previously upon application of moisturizers in several instances (2,6,5,19). The increase in skin capacitance and conductivity upon increased hydration resulting from application of moisturizers has been studied previously (1). Humectants such as glycerin, pyrollidine carboxylic acid, or urea in o/w emulsions, in comparison to petro- leum jelly have proven to be effective in increasing skin capacitance in normal adult forearm skin (20). Matsumoto et al. in 2007 reported an increase in skin capacitance in infantile leg sites upon application of an o/w cream containing glycoceramide, while heparinoid-containing creams were more effective in the chest regions (3). While this variability in effectiveness with the site of application may be explained to be a result of the absorbtion of humectants by the skin, in the case of emollients like HP or CCT, due to their high molecular weight, we may speculate that the increase in capacitance is a result of the formation of an epicutaneous fi lm rather than absorbtion. However, further studies on skin absorption and penetration will help to ascertain this hypothesis. ACKNOWLEDGMENTS The fi nancial support from INEOS Oligomers to carry out this work is gratefully acknowledged. We thank Dr. Stephen M. Greenberg for critically reviewing this manu- script and for his valuable comments. Figure 5. Polarized photographs taken by View Charm: baseline (upper row), untreated, versus 6-h post-treatment (lower row).
JOURNAL OF COSMETIC SCIENCE 24 REFERENCES (1) L. G. Grove, The effect of moisturizers on skin surface hydration as measured in vivo by electrical con- ductivity, Curr. Ther. Res., 50(5), 712–719 (1991). (2) M. Lodén, Barrier recovery and infl uence of irritant stimuli in skin treated with a moisturizing cream, Contact Dermatitis, 36(5), 256–260 (1997). (3) T. Matsumoto, H. Yuasa, R. Kai, H. Ueda, S. Ogura, and Y. Honda, Skin capacitance in normal and atopic infants, and effects of moisturizers on atopic skin, J. Dermatol., 34(7), 447–450 (2007). (4) I. Buraczewska, B. Berne, M. Lindberg, H. Törmä, and M. Lodén, Changes in skin barrier function following long-term treatment with moisturizers, a randomized controlled trial, Br. J. Dermatol., 156(3), 492–498 (2007). (5) I. Buraczewska, U. Broström, and M. Lodén, Artifi cial reduction in transepidermal water loss improves skin barrier function, Br. J. Dermatol., 157(1), 82–86 (2007). (6) M. Lodén, Role of topical emollients and moisturizers in the treatment of dry skin barrier disorders, Am. J. Clin. Dermatol., 4(11), 771–788 (2003). (7) M. Lodén, Biophysical properties of dry atopic and normal skin with special reference to effects of skin care products, Acta Derm. Venereol. (Stockh.), 192(Suppl), 1–48 (1995). (8) T. W. Fischer, W. Wigger-Alberti, and P. Elsner, Assessment of “dry skin”: Current engineering meth- ods and test designs, Skin Pharmacol. Appl. Skin Physiol., 14(4), 183–195 (2001). (9) T. W. Fischer, W. Wigger-Alberti, and P. Elsner, Direct and non-direct measurement techniques for analysis of skin surface topography, Skin Pharmacol. Appl. Skin Physiol., 12(1–2), 1–11 (1999). (10) I. Afriat and D. Gagnebien, Use of polydimethylsiloxane containing glucoside groups as a moisturizing agent in a consmetic or dermatological composition. L’Oreal Patent 6066326, May 23rd, 2000. (11) I. Travkina, M. Raouf, and H. E. Pahlck, Wear resistant cosmetic formulation, Avon Products Patent 6309629, October, 2001. (12) K. A. Traul, A. Driedger, D. L. Ingle, and D. Nakhasi, Review of the toxicologic properties of medium- chain triglycerides, Food Chem. Toxicol., 38(1), 79–98 (2000). (13) F. Q. Hu, Y. Zhang, Y. Z. Du, and H. Yuan, Nimodipine loaded lipid nanospheres prepared by solvent diffusion method in a drug saturated aqueous system, Int. J. Pharm., 348(1–2), 146–152 (2008). (14) International Federation of the Society of Cosmetic Chemists, Principles of Product Evaluation: Objective Sensory Methods, Euro 21,40 (1987). (15) D. D. Verma, S. Verma, G. Blume, and A. Fahr, Particle size of liposomes infl uences dermal delivery of substances into skin, Int. J. Pharm., 258(1–2), 141–151 (2003). (16) P. H. Hoet, I. Brüske-Hohlfeld, and O. V. Salata, Nanoparticles—Known and unknown health risks. J. Nanobiotechnol., 2(1), 12 (2004). (17) E. Yilmaz and H. H. Borchert, Design of a phytosphingosine-containing, positively-charged nanoemul- sion as a colloidal carrier system for dermal application of ceramides, Eur. J. Pharm. Biopharm., 60(1), 91–98 (2005). (18) E. Yilmaz and H. H. Borchert, Effect of lipid-containing, positively charged nanoemulsions on skin hydration, elasticity and erythema—An in vivo study. Int. J. Pharm., 307(2), 232–238 (2006). (19) K. Endo, N. Suzuki, O. Yoshida, H. Sato, and Y. Fujikura, The barrier component and the driving force component of transepidermal water loss and their application to skin irritant tests, Skin Res. Technol., 13(4), 425–435 (2007). (20) M. Lodén and M. Lindberg, The infl uence of single application of different moisturizers on skin capacitance, Acta Derm. Venereol. (Stockh.), 71, 79–82 (1991).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)