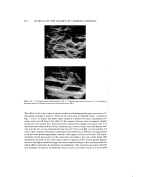
WATER-IN-OIL EMULSIONS 287 EXPERIMENTAL GEL FORMATION OBTAINED BETWEEN SURFACTANTS AND AMINO ACIDS Materials: Most of the surfactants used in this study were commercially available. For example, Sunsoft © O-30B* (glycerol monooleate) was used as a standard surfactant for many of the experiments. Whenever necessary, those synthesized in the usual way or those fractionated by molecular distillation were used. The amino acids and their salts were of special reagent grade. Other reagents used were also of the same grade. Distilled water was used throughout the study. METHOD a. Gel Formation.' Surfactants and amino acids or their salts, which were possible to form the gels, were classified by the following simple method. The surfactant was added to an aqueous solution of the amino acid or its salt at room temperature and stirred with a laboratory mixer. A gel was formed as shown in Fig. 1. All of the gels obtained by this method were observed as to their electrical conductivity and their stability in hot water. Figure 2 illustrates this property and only those without electrical conductivity and insoluble in water were selected for further study. b. Other Measurements.' In order to investigate the various functions of the gels, the chemical structure of the surfactants, the structure and properties of the gels obtained, and the effects of amino acids, etc., were examined by the following methods: Figure 1. Example of gels prepared between surfactants and aqueous solution ofamino acids *Taiyo Kagaku Co., Ltd., 62 Akahori, Yokkaichi, Mie, Japan.
288 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS :•.•:•.-.•.•'•:?i•}=• 5 "• •'7- -'? _."-?.•.'• :'¾•"'•,5:i '•,•, "•5•,5•::i{=• '•'. --•.=--•k:5%•-,.7 •-•.' '% • • 5 .... ?•.•.5?•ei::•:•?'?'• "-'•:•'•'.::•,:•5•¾:-": ..•'-•'•,i. ?-? •': -,.'P%: ? ?•.-•: '••:•%•5•%',,s'•'•" •'"'•x• •.•. • Figure 2. Stability of gels in hot water: (a) stable gel (b) unstable 8el (1) X-Ray Analysis.' Two types of X-ray diffractometers, Rotaflex* and JRX-12VA•- were used to elucidate the structures of the surfactants and gels by means of small angle scanning and can/era method at 5 C Targe.t Cu. 2 ø (2) Differential Thermal Analysis (DTA).' The gel sealed in aluminum cell was measured at 0 to 70øC, raising the temperature 2.5øC/rain by a scanning type DSC.* (3) NMR.. Variation of chemical shift with proton in water of the aqueous solution to which various solutes were added, was measured by Hitachi$ R-20 type NMR at 34øC. (4) Phase Inversion Temperature (PIT).' Influence of the addition of amino acids on the PIT was measured with Squalane•Beeswax•POE(6) oleyl alcohol ether 5 per cent (w/w)•water system (volume ratio = 0.6). (5) Heat of Solution.. A sealed ampiale containing about 2 g of the surfactant, Sunsoft O-30B, was broken in 50 g aqueous solution of a.Nino acid having various concentra- tions. of heat on mixing (cal/g) was me'asured by a twin type microcal- orimeter** at 35øC.
Evolution (6) Water Content Migration to the Surfactant Phase.' The amount of water migrating into the surfactant (Sunsoft O-30B) through an interface of the surfactant and an *Rigaku Denki 8-2, Kandasurugadai, Chiyodaku, Tokyo, Japan. -•Japan Electron Optics Laboratory, 1418, Nakagami, Akishimashi, Tokyo, Japan •2 Shibanishikubo, Minatoku, Tokyo, Japan. **Applied Electric Laboratory, 4-672 Sekimachi, Nerimaku, Tokyo, Japan.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)