REFRACTIVE INDEX MATCHING Table II Clear Eye-Moisturizing Gel Ingredient Silicone oil phase Cyclomethicone and dimethicone (DC 1501, Dow Corning) Cyclomethicone and dimethicone copolyol (DC 5225, Dow Corning) Cyclomethicone (DC 344, Dow Corning) AHA water phase Water, deionized Glycerin Glydant Glycolic acid and arginine (AHCare G-60, Cognis) Rloil = (10 X 1.397 + 10 X 1.398 + 5 X 1.394)/25 = 1.3967. R/wctter = (35.5 X 1.333 + 27 X 1.468 + 12.5 X 1.428)/75 = 1.3966. Table III Weight % 10.0 10.0 5.0 36.1 26.25 0.15 12.5 100.0 Clear Silicone Styling Gel Calculated for Both Oil and Water Phases Ingredient Silicone oil phase Cyclomethicone (Rhodorsil 45V5, Rhodia) Polyacrylamide, C13-14 isoparaffin, and laureth-7 (Sepigel 305, Seppic) Cyclomethicone, phenyltrimethicone, and dimethicone (Gelaid 5565, Chemsil) PVP!VA water phase Water, deionized Glycerin PVP/V A copolymer solution (50% active) (Luviskol VA 73W, BASF) DMDM hydantoin (Glydant, Lonza Group) R/oil = (4 X l.396 + 3 X 1.446 + 20 X 1.402)/27 = 1.4060 R/uwer = (26.5 X 1.333 + 21.35 X 1.472 + 25 X 1.427 + 0.15 X 1.425)/73 = 1.4060 Weight % 4.00 3.00 20.00 26.50 21.35 25.00 0.15 100.00 259 RI value 1.3971 1.3975 1.3942 1.333 1.468 1.425 1.428 RI value 1.396 1.446 1.402 1.333 1.472 1.427 1.425 similar (in terms of structure and spatial filling) to one another that molecules of one component can replace molecules of another component in the solution without chang ing the solution's energy or spatial structure. In ideally dilute solutions, all solutes are present in very low concentrations and the solvent weight percentage approaches 100%. In cosmetic formulations, solutions are neither ideal nor ideally dilute because the solvent and solutes cannot be very similar, and the concentrations of solutes cannot be impractically low. They are non-ideal solutions containing both electrolytes and non electrolytes. More precise calculations would require chemical potential (µ), solute activity (a), and activity coefficients (-yJ The calculation becomes very complicated and impractical for cosmetic chemists. Fortunately, a simplified calculation is sufficient to serve the purpose of designing formulas. In the process of making the oil phase and water phase, for example, the RI will deviate slightly from the calculated value. The deviation in the oil phase is usually very small because all the RI values of the oil-phase ingredients are very close. Consistent RI
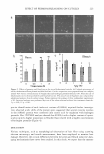
260 JOURNAL OF COSMETIC SCIENCE readings of oil fluids result from the similarity in both the molecular structure and the interaction between these molecules. However, there is a noticeable RI deviation in water-phase preparation. In matching RI values of water and oil phases, one uses water (low RI value) and glycols (high RI values). In addition, the molecular interaction between glycol and water is different from the interaction between those molecules by themselves. Therefore, the formula needs further refinement to assure proper index matching. Various glycols are used to raise the refractive index of the aqueous phase to match that of silicone oil (RI - 1.4) or isoparaffin (RI - 1.43 ). The refractive index values for several glycols as a function of concentration are shown in Figure 2 for a two-component system. The glycol aqueous concentrations vary from 10% to 80%. Different deviations are seen in the plot. Glycerin shows negative deviation and three glycols show positive deviation. Hexylene glycol shows the largest positive deviation. Glycereth-7 shows very little deviation. All deviations are concentration-dependent. Deviations are compared with the calculated values shown in Figures 3-5. Negative deviation is observed for glycerin in aqueous solution (Figure 3). The largest negative deviation is seen for glycerin at the concentration of 30%, where there is a -0.51 % deviation (1.367-1.37395 == -0.00695) from the calculated value. 1.48 1.44 � Cl) 1.4 -----+-- Propyl G Cl) -a-Butyl G 1.36 �Hexyl G -e-Glycerin �?l(-G lycereth-7 1.32 0 20 40 60 80 100 Percentage of Glycol Figure 2. Concentration-dependent RI values of several glycol aqueous solutions.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)