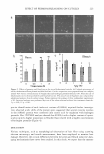
REFRACTIVE INDEX MATCHING 261 1.48 1.44 -� 1.4 i:: � � 1.36 • RI Glycr Ext RI Glycr Cal 1.32 0 20 40 60 80 100 Percentage of Glycerin Figure 3. Theoretical and experimental concentration-dependent RI values of glycerin aqueous solution. Solid line is experimental and dashed line is theoretical. Positive deviation is seen for hexylene glycol aqueous solution in Figure 4. It is the largest positive deviation among all the studied glycols (Figure 2). The experimental RI value of hexylene glycol is even larger than the value of glycerin solution at concentra tions in the range of 10-35%. The largest deviation of the refractive index for hexylene glycol is at the concentration of 60%, where it deviates 0.97% from the calculated value. Figure 5 demonstrates slightly negative deviation at lower concentration and slightly positive deviation at higher concentration for glycereth-7 aqueous solution. Glycereth-7 is a unique case since deviations run from slightly negative at low concentration (below 40%) to slightly positive at high concentration (above 50%). Negative deviation at lower concentration is comparable to what is observed in aqueous glycerin, and positive deviation at higher concentration is comparable to that of hexylene glycol. As a way of explaining deviation, the specific gravities of aqueous solutions of glycerin and three glycols (propylene glycol, butylene glycol, and hexylene glycol) at 50% by weight were measured and are listed in Table IV. The specific gravity of glycerin solution is lower than the calculated value (average of two components), which indicates a slight increase in volume after glycerin and water are mixed. The volume increase might be related to the difference in intermolecular interactions between water mol-
262 1.44 1.42 t1 � 1.4 � � 1.38 ;.. ·-= � 1.36 1.34 1.32 o JOURNAL OF COSMETIC SCIENCE 20 40 ----■� RI Hexylg Ext RI Hexylg Cal 60 80 Percentage of Hexylene Glycol 100 Figure 4. Theoretical and experimental concentration-dependent RI values of hexylene glycol aqueous solution. Solid line is experimental and dashed line is theoretical. ecules, between glycerin molecules, and also between water and glycerin molecules. The interaction between water molecules and between glycerin molecules is possibly stronger than the interaction between water and glycerin molecules. A decrease in specific gravity results in a decrease in optical density, which is observed as a negative deviation in refractive index measurements. It turns out that glycol solutions behave differently compared to glycerin solutions, as is illustrated in Table IV. The observed increase in specific gravity corresponds to a decrease in volume after glycols dissolve in water. A plausible explanation could relate to spatial filling, since the three glycol molecules are structurally larger than water molecules. Also, water molecules can fill voids between glycol molecules in glycol solution. An increase in solution specific gravity leads to an increase in optical density, which results in positive deviation in refractive index measurements. Propylene glycol, butylene glycol, and hexylene glycol all show positive deviation (Fig ure 2). However, propylene glycol demonstrates the least and hexylene glycol the most. The nature of deviation is related to molecular interaction between water molecules, between glycol molecules, and between water and glycol molecules. These three glycols have the same diol (dihydroxy) functional group on two carbon atoms (illustrated in
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)