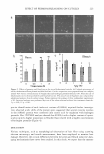
1.48 1.44 � 1.4 ·-= "' e � � 1.36 1.32 0 REFRACTIVE INDEX MATCHING 20 40 -•- Glyrth-7 Exp Glyrth-7 Cal 60 80 Percentage of glycereth-7 263 100 Figure 5. Theoretical and experimental concentration-dependent RI values of glycereth-7 aqueous solution. Solid line is experimental and dashed line is theoretical. Glycol Glycerin Hexylene glycol Butylene glycol Propylene glycol Table IV Specific Gravity of Glycols in 50% Aqueous Solution SPG Exp SPG Cal 1.1268 1.1304 0.9870 0.9613 1.0251 1.0085 1.0354 1.0184 �SPG -0.0036 +0.0257 +0.0166 +0.0170 Figure 6). The more carbon contained within the molecular structure, the bulkier the glycol molecule. The more carbon the molecule has, the greater the difference will be in molecular interaction between glycol and water molecules. The greater this difference, the more positive is the observed deviation (Figure 2). Hexylene glycol shows the greatest increase in specific gravity and a concomitant positive deviation in the refractive index. Negative RI deviation corresponds to a decrease in the specific gravity of glycerin solution. Positive RI deviation corresponds to an increase in the specific gravity of glycol solution. Based on these observations, it is reasonable to generalize that any
264 JOURNAL OF COSMETIC SCIENCE glycerin propylene glycol butylene glycol hexylene glycol Figure 6. Molecular structures of glycerin, propylene glycol, bucylene glycol, and hexylene glycol. increase or decrease in specific gravity leads to an increase or decrease in optical density, which results in RI values that deviate positively or negatively from calculated values, respectively. The dominant interaction forces between molecules of water and glycols or glycerin are hydrogen bonds since all of them are polar molecules with hydroxyl groups. Hydrogen bonds play important roles for the decrease in specific gravity in glycerin solution. The reason for volume increase (specific gravity decrease) when forming glycerin aqueous solution is that there is stronger hydrogen bonding between glycerin molecules than the hydrogen bonding between molecules of water and glycerin. That the process accom panies a significant heat release when hydration takes place also indicates the different strength of hydrogen bonding. Glycols, however, are hydrated differently. That there is a volume decrease (specific gravity increase) when forming glycol solutions indicates that there is weaker hydrogen bonding between glycol molecules than the hydrogen bonding between molecules of water and glycols. It is apparent that the most efficient way to raise the refractive index of the aqueous phase is to use a combination of hexylene glycol in the range of 20-30% with either glycerin, glycereth-7, or butylene glycol. Propylene glycol is less efficient at raising the RI of the water phase because it has the least positive deviation and the lowest refractive
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)