JOURNAL OF COSMETIC SCIENCE 34 area of 50 × 50 μm was randomly rastered by primary ions and was charge-compensated by low-energy electron fl ooding. The amount of 18-MEA adsorbed to the hair fi ber was measured using liquid chromatog- raphy/mass spectrometry (LC-MS, Agilent Technologies, Palo Alto, CA). Hair fi bers were immersed in chloroform/methanol (1:1 by volume) for 1 h at room temperature. The ex- tracts were dried using a nitrogen stream. The residues were then dissolved in chloroform/ methanol (1:9 by volume). In the system, a 1100 binary pump was connected to two mo- bile phases [M1, methanol/water (8:2 by volume) containing 100 mM ammonium acetate and 50 mM acetic acid and M2, methanol containing 100 mM ammonium acetate and 50 mM acetic acid] that were eluted at a fl ow rate of 0.2 ml/min. The mobile phases were pro- grammed consecutively, as follows: a linear gradient of M1 100–0% (M2 0–100%) be- tween 0 and 20 min, an isocratic elution of M1 0% (M2 100%) for 10 min, and an isocratic elution of M1 100% (M2 0%) from 30.1 to 40 min for column equilibrium (a total run time of 40 min). The injection volume was 5 μl. The column (L-column ODS 2.1-mm inner diameter × 150 mm Chemicals Evaluation and Research Institute, Tokyo, Japan) temperature was maintained at 40°C. ESI measurements in the mass spectrometer were performed with the following settings: polarity, negative heater temperature of nitrogen gas, 350°C fl ow of heated dry nitrogen gas, 11.01/min nebulizer gas pressure, 30 psi capillary voltage, -4000 V fragmenter voltage, 200 V. The selected ion monitoring (SIM) measurement in negative ion ESI was performed using the unit mass resolution mode. To detect deprotonated ions for 18-MEA, m/z = 325.2 was monitored. Measurement of surface properties of mica (A) Atomic force microscopy AFM images of the adsorbed layers on the mica surfaces were obtained using a Nanoscope IIIa Multi Mode AFM (Veeco Instruments, Santa Barbara, CA) with E-scanner. Tapping mode imaging was used to obtain the topography images of the absorbed membrane layers. To ensure that imaging the membrane caused no damage, the tapping force was set at the lowest possible level. The nominal spring constants of cantilevers are reported by the manufacturer to be 20-100 N/m, respectively. All images presented in this work were obtained reproduc- ibly over at least three spots on the sample surfaces. The images were acquired with a scan rate of either 0.5 or 1.0 Hz and were fl attened with a fi rst-order polynomial before analysis. The mechanical properties of the adsorbed membrane were analyzed by the AFM scratch- ing method. Scratching the adsorbed membrane was performed in contact mode with constant force, and a micro-fabricated tip made of silicon nitrate and a cantilever having a spring constant of 0.38N/m were used. First, an image (typically 5 μm × 5 μm) of the absorbed membrane was acquired then a smaller area (typically 1 μm × 1 μm) was scanned while loading the hard tip onto the surface. Following this, the scanning was repeated over the larger area. This method is hereafter called “scratching.” Thus, if the absorbed layer was strongly bound to the surface, it was harder to remove. (B) Angle-resolved X-ray photoelectron spectroscopy (ARXPS) ARXPS is a nondestructive method to obtain elemental and chemical-state information as a function of depth. ARXPS data was obtained using a Quantera SXM spectrometer (ULVAC PHI, Kanagawa) with a monochromatized Al K alpha X-ray source at 15 kV and 25 W. Survey spectra were recorded at a takeoff angle of 45° with pass energy of 280 eV by a cylindrical-mirror analyzer. Angle-resolved spectra were recorded at fi ve takeoff
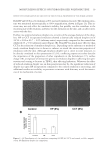
18-MEA DEPOSITION ON HAIR 35 angles, 70, 45, 25, 15, and 10 degrees, in a high-energy resolution mode with pass energy of 112 eV. RESULTS CHARACTERIZATION OF ALKALINE-COLOR-TREATED WEATHERED HAIR Surface properties of alkaline-color-treated weathered hair are shown in Figure 1. Since 18-MEA is covalently bound to the cuticle surface via a thioester linkage, it can be re- moved with alkaline treatments such as perming, coloring, and bleaching. It is well known that the reduction of 18-MEA affects surface properties of hair such as hydrophobicity and friction. Figure 1(A) shows the relative ion yield of 18-MEA versus the total ion yield on the outermost surface of hair measured by TOF-SIMS. In the case of alkaline-color-treated weathered hair, most of the 18-MEA has been removed. Figure 1(B) shows the hydropho- bic property of normal hair and alkaline-color-treated weathered hair. A higher value means that the surface is more hydrophobic, and a lower value means that the surface is more hydrophilic. The contact angle of normal hair was around 90°, which means that it was hydrophobic because of the presence of 18-MEA. On the other hand, that of alkaline- color-treated weathered hair was around 65°, which means that it was more hydrophilic because of the absence of 18-MEA and the oxidation of sulfur groups to sulfate. Figure 1(C) shows the dynamic friction coeffi cients of normal hair and alkaline-color-treated weathered hair. It shows that surface friction increased when 18-MEA was removed. Since it was confi rmed that the results obtained here correspond to previous knowledge, alkaline-color-treated weathered hair was used in the subsequent experiments. The objective for this study was to attach 18-MEA onto alkaline-color-treated weathered hair and to provide persistent hydrophobicity and low friction on an alkaline-color-treated weathered hair surface. In the following sections, contact angles after one instance of shampooing were measured in order to make sure that the damaged hair surface main- tained its hydrophobicity even after shampooing. Figure 1. Surface properties of alkaline-color-treated weathered hair. Relative ion yield of 18-MEA versus the total ion yield (A), contact angle (B), and dynamic friction coeffi cient (C) are presented. The bars repre- sent means for n=5 the whiskers represent the standard deviations. (a) Normal hair. (b) Alkaline-color- treated weathered hair.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)