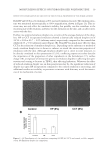
JOURNAL OF COSMETIC SCIENCE 40 18-MEA/DAPS (b) or 18-MEA/STAC (c), a square groove was observed. On the other hand, treatment by 18-MEA/SPDA (a) showed no such groove. These results indicate that the 18-MEA/SPDA conditioner adsorbed homogeneously on the mica surface and had a high wear resistance. Regarding the thickness of the layer, it was estimated that the layer of 18-MEA/SPDA was about 1-nm thick. Molecular orientations of the adsorbed layers investigated by ARXPS are shown in Figure 11. The peak area ratio of COO (carboxyl) from 18-MEA/C (carbon) from conditioner ingredients, such as surfactant, stearyl alcohol and so on, increased with the measurement depth. The peak area ratio of N (nitrogen)/C (carbon) from SPDA increased as a function of the measurement depth in a similar manner. These results suggest that the hydrophilic moiety of the molecules, the carboxyl group of 18-MEA and the amide group of SPDA, attach to the mica surface and orient the alkyl chain (the hydrophobic moiety) to the air interface. Taking into account our results obtained here, the thickness of 18-MEA/SPDA Figure 9. AFM height images of adsorbed layer on mica surface. Dark areas are mica without sorption bright areas are sorbed layer from conditioner. (a) 18-MEA/SPDA. (b) 18-MEA/DAPS. (c) 18-MEA/STAC. Figure 10. AFM height images of adsorbed layer on mica surface. Dark areas are mica without sorbed com- pound bright areas are sorbed chemicals from conditioner. The white squares indicate where 1000 nm ×1000 nm scratching tests were done by rastering the tip with constant force. (a) 18-MEA/SPDA. (b) 18-MEA/ DAPS. (c) 18-MEA/STAC.
18-MEA DEPOSITION ON HAIR 41 is about 1 nm, and since the alkyl chain length of 18-MEA is 2.39 nm, the 18-MEA/ SPDA layer orients at an angle of around 25° to the air interface. Thus, the following model (Figure 12) is suggested, based on the results obtained from the present investigation: 18-MEA/SPDA forms a layer 1 nm in thickness, with both 18-MEA and SPDA bound tightly to the surface by the carbonyl and amide groups, orienting the hydrophobic part to the air interface at an angle of around 25°. Another question was whether SPDA is special for providing persistent hydrophobicity to alkaline-color-treated weathered hair surfaces. Additional studies, on the dynamic contact angles and the amount of 18-MEA sorption measurements, were conducted to elucidate the issue with the complexes of 18-MEA and a variety of long-chain tertiary amines. Table III shows the dynamic contact angles and the amount of 18-MEA sorption of alkaline-color-treated weathered hair treated with 18-MEA/long-chain tertiary amines. The chemical structures of these tertiary amines and the logP value, which is calculated by the ClogP method, are also listed in Table III. The combination of 18-MEA with SPDA or DSDA made the alkaline-color-treated weathered hair surfaces hydropho- bic, and their hydrophobicity was maintained even after one instance of shampooing with a plain shampoo [15 wt% of sodium polyoxyethylene lauryl ether sulfate (2.5 E.O.) with 2 wt% N,N-bis(2-hydroxyethyl)-dodecanamide solution adjusted to pH7 with phosphoric acid], while the combination of 18-MEA with SHDA and DAPS could not. Figure 11. Molecular orientation analysis of 18-MEA/SPDA membrane by angle-resolved X-ray photoelec- tron spectroscopy (ARXPS). θ represents photoelectron take-off angle. (a) Angle dependence of nitrogen/ carbon concentration derived from SPDA. (b) Angle dependence of COO/C-C concentration derived from 18-MEA. Figure 12. Schematic diagram of the cuticle surface of alkaline-color-treated weathered hair treated with 18-MEA/SPDA. 18-MEA/SPDA forms a layer with high wear resistance, orienting the alkyl chain (hydro- phobic moiety) at an angle of around 25° to the air interface.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)