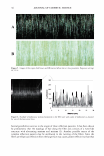
96 JOURNAL OF COSMETIC SCIENCE LIGHT, RADIATION, AND ENERGY Exposure to radiation is a cause of concern to the personal care chemist, and in particular the radiation that has a great amount of energy, like UV, is known to cause many undesirable effects. For example, dyes, fragrances and carbomer-type thickeners exhibit varying degrees of instability upon exposure to light. This article explores the potential for structural damage caused by UV radiation to human hair. Appendix I shows a number of basic equations that define the relationship of quantities and units of measures commonly used to describe light, radiation, and wavelength Appendix II is a summary table of more complex functions like irradiance, radiant power, and radiant energy. The following discussion will consider, light and its energy, chemical bonds and the energy required to break them, and the radiant energy coming from the sun that reaches the earth's surface and the spectral distribution of such energy. Table I shows the wavelength, frequency, and energy of different radiation. Table II shows the energy required to break chemical bonds (bond dissociation energy) it is evident that the energy associated with UV is sufficient to break chemical bonds within the organic substrates (proteins, keratin, hair) in addition, the radiation of such energy as UV is capable of inducing free radical reactions that would have a lower activation energy and be even more likely than bond cleavage. The generally accepted mechanism for photo-oxidation is well presented by Robbins (2). Figure 1 shows the actual values of the radiation power reaching the earth's surface at a specific point in time and geographical area. A large variation in the spectral power distribution is to be expected between times of the year, hours of the day, and the geographic location at which the measurements are taken (3 ). The unit of measure of radiant power received per unit surface area is irradiance (W m- 2 ). Ir radiance is often associated with a wavelength or wavelength range (W m - 2 nm - 1 ) for instance, an average 1.5 W m- 2 nm- 1 over a wavelength range of 200 nm would give a total 300 W m- 2 total irradiance. The summation of irradiance over a period of time is called irradiation energy (or radiant exposure) e.g., irradiance of 1. 5 W m- 2 over a period of five hours(= 18,000 s) is equal to 27,000 J m- 2 . A radiant exposure of 3-6 KWh m- 2 day- 1 (= 10.8 x 106 - 21.6 x 106 J m- 2 day- 1 ) is the average for the US during the months of July and August (6). In comparison, in Australia, the global solar exposure in August is between 13 and 22 x 106 J m- 2 day- 1 (6). The values reported above refer to the total solar radiation intensity, visible (VIS), and UV and infrared (IR) rays. Table III gives a breakdown of the ratio between VIS and UV radiation. As seen in Table III, the irradiance ratio between UV and VIS radiation is approximately 1 to 10. Generally speaking, when the measurement of the solar radiation is referred to as total} that means that it includes visible, ultraviolet, and infrared (VIS, UV, IR) rays when it is referred to as global} it means that the measurement was carried out in such a way as to capture the radiation from the sky, avoiding the radiation resulting from reflection on the ground. When the total radiation is considered, the relative proportions of UV, VIS, and IR, respectively, are 4-6%, up to 52%, and up to 42%.
Table I Radiation's Frequency and Wavelength (1): Planck's Equation* Applies, and It Describes the Indirect Proportionality Between the Wavelength of the Radiation** and the Energy That Is Associated With It (the longer the wavelength, the lower the energy) Wavelength, A (m) 10- 12 - 10-10 10-10 - 10-3 (= 10 nm) 10- 3 (10 nm) - 3.8 x 10- 7 (380 nm) 3.8 x 10-7 (380 nm) -7.8 x 10-7 (780 nm) 7.8 X 10-7 - 10-4 10- 4 - 10-2 10-2 - ... * See Appendix I. Radiation )'-rays X-rays UV VIS Infrared Microwaves Radio waves Frequency, v (Hz or s- 1 ) 3 X 1020 - 3 X 10 18 3 X 10 18 - 3 X 10 16 3 X 10 16 - 7.8 X 10 1 4 7 .8 X 10 1 4 - 3.8 X 10 1 4 3.8 X 10 1 4 3 X 10 1 2 - 3 X lQ lO 3 X 10 1 0 - ... Energy of 1 mol of photons* (KJ mol- 1 ) 11.95 X 107 -11.95 X 105 11.95 X 105 - 11.95 X 103 11.95 X 103 - 314 314 - 153 153 - 1.19 1.19 - 1.19 X 10-2 1.19 X 10-2 - •.. ** Very often, the UV radiation is referred to as "UV light" this is strictly speaking inaccurate, as the definition of light implies that it is visible to the human eye. UV radiation is not visible therefore, it is a radiation rather than a light. m 1-Tj 1-Tj m n ,.., 0 C � u ,.., 5 0 z ::r: � ,.., � C n ,.., C � m \0 ---J
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)