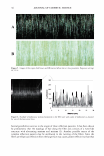
TARGETED DELIVERY OF SALICYLIC ACID 77 5 4.5 N 4 E 3.5 3 ::I c 2.5 2 Q) 1.5 Q) 1 0.5 o�----- Formula *Forrnu1ations are in table I B. Penetration was measured at 24 hours -Anionic system is 0.5% sa1icylic acid reference forrnu1ation without polymer -0% polymer - formula 9 is the nonionic isoceteth-20 system with 0.5% SA and 0% polymer -1 % polymer - formula 10 is the nonionic isoceteth-20 system with 0.5% SA and 1 % polymer Figure 6. Penetration of 0.5% salicylic acid into the receptor: Effect of polymer dose and comparison to reference products. Table IV 14-Day Cumulative Irritation Potential of Several Hydroalcoholic Solutionsa Hydroalcoholic acne treatment formulations6 2% Salicylic acid 0% polyolprepolymer nonionic isoceteth-20 (formulation 1) 2% Salicylic acid 2% polyolprepolymer nonionic isoceteth-20 (formulation 3) 2% Salicylic acid 3% polyolprepolymer nonionic isoceteth-20 (formulation 4) 0.5% Salicylic acid 0% polyolprepolymer nonionic isoceteth-20 (formulation 9) 0.5% Salicylic acid 1 % polyolprepolymer nonionic isoceteth-20 (formulation 10) 0.5% Salicylic acid commercial anionic formulation 2% Salicylic acid commercial anionic formulation Test control (water/sodium chloride) a 14-day patch test sum of daily irritation scores. 6 Formulations in Tables IA or IB. c Statistical differences in test samples are discussed in the text. DISCUSSION Irritation scorec 223 182 193 88 73 338 Extreme irritation, pulled off test 24 The hydroalcoholic formulations studied contain a very mild nonionic surfactant, iso ceteth-20. Nonionic surfactants are perhaps the mildest of all surfactant systems to human skin (17 ,18), and our irritation studies reported herein support this finding. Like other surfactants, they also micellize (self-associate) in solution at a low concentration and can trap salicylic acid in their micellar or vesicular reservoir. (The micellar or vesicular reservoir may be formed on the skin surface after alcohol evaporation, as the presence of alcohol in the solution may not allow micellization to occur.) As a result, salicylic acid will penetrate more slowly into and through the skin rather than as a large
78 JOURNAL OF COSMETIC SCIENCE dose after the initial exposure. These formulations also incorporate the nonionic poly urethane polymer, polyolprepolymer-15. Both of these excipients tend to associate with the salicylic acid drug via weak hydrophobic bonds and alter the "activity" of the drug and the penetration profile of the drug into and through the skin. This results in slow release of the drug and targeted delivery into the epidermis rather than through the skin. Reduction of the initial rapid rate of delivery of the drug into the receptor parallels the reduction of irritation found in the cumulative irritation test. However, even the nonionic hydroalcoholic formulations without the polymer are milder than the marketed reference formulations containing anionic surfactants. Because of the absence of a charge in the nonionic surfactants, they do not dissociate/denature the stratum corneum as readily as certain anionics (17,18). This confirms that the surfactant type is the key factor dictating the enhanced mildness of the surfactant systems and suggests that the increased micellar or vesicular reservoir characteristic of systems with a lower critical micelle concentration (as is the case of these nonionic versus anionic surfactants) may explain in part the reduced irritancy of the system. This presumably occurs by greater entrapment of salicylic acid in the micellar reservoir and by minimiz ing the surfactant monomer available to attack the skin barrier, since most of the surfactant is tied up in the micelle (17,18). Arguably, the surfactant micellization or vesicle formation may occur on the skin surface after the alcohol has evaporated. The importance of surfactant type is further substantiated by the fact that a reference (com mercial) anionic formulation with 0.5% salicylic acid was substantially more irritating than any nonionic surfactant system with 2% salicylic acid. Yet the anionic formulation with 0.5% salicylic acid delivered the least amount of salicylic acid of the two (com paring Figure 6 with the other figures, for the 2% nonionic formulations, although comparing results between different experiments is not entirely reliable). Thus the surfactant type rather than the concentration of salicylic acid is the predominant factor controlling irritancy. One other explanation for the findings is in order. It is possible that the predominant effect of slowing down delivery of salicylic acid by the surfactant systems could be due to competition for binding surfaces on the stratum corneum. Such surfaces are hydro phobic in nature, a fact that is well published, and the hydrophobic part of the surfac tants could adsorb to these regions of the keratin, thus preventing the salicylic acid from binding to these same sites. However, the relationship between binding to keratin and penetration of salicylic acid is unclear. An interesting experiment would be to assess the relationship of salicylic acid delivery to the dose of the surfactant both above and below the critical micelle concentration of the surfactant. It would also be interesting to isolate micelles or vesicles after alcohol evaporation and determine the extent of entrapment of salicylic acid into these structures [see review by Rhein (17 ,18)}. Alcohol could also be a contributor to the irritancy. However, the findings suggest this is not the case. The low-alcohol (18%) commercial formulations with 0.5% salicylic acid in an anionic surfactant system were more irritating than the 42% alcohol formulations containing 2% salicylic acid and the nonionic surfactant. Alcohol does not seem to play a major role in controlling the irritation except for the initial solubilization of the salicylic acid. The learnings from these studies are that polyolprepolymer-15 and isoceteth-20 can be used to target the delivery of salicylic acid preferentially to the epidermis and diminish the rate of penetration through the skin into the receptor, which would be the blood-
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)