JOURNAL OF COSMETIC SCIENCE 166 lipid nanoparticles (SLNs), a colloidal delivery system, consist of a high melting point solid lipid forming a lipid matrix surrounded with surfactant (5,8). SLNs have several advantages including high tolerability, rapid biodegradation, high bioavailability, high drug-loading capacity, good production scalability, and a lack of organic solvents in the preparation process. SLNs can also provide a controlled release profi le for many active agents and increase the skin penetration rates of the active agents (3,8). Homogenization, solvent emulsifi cation or evaporation, ultrasonication, and solvent diffusion methods are used to prepare SLNs (9). The aim of the present study was to prepare SLNs containing Caf with controlled release and to evaluate the formulations with in vitro experiments. MATERIALS AND METHODS MATERIALS Softisan 100® (Hydrogenated Coco-Glycerides) was obtained from Cremer Oleo GmbH & Co. KG (Hamburg, Germany). Tween 20® (Polysorbate 20), Span 20® (Sorbitan Laurate), and Caf were purchased from Merck KGaA (Darmstadt, Germany). All organic solvents and other chemicals were analytical grade and obtained from Merck KGaA. PREPARATION METHOD FOR CAF-SLNS Caf was dissolved in 80°C water. The aqueous solution was added into the oil phase which comprised a Softisan 100® and Span 20® mixture at the same temperature. It was mixed under constant stirring (27,000 rpm) with a Silent Crusher M homogenizer (Heidolph®, Schwabach Germany) at 80°C for 2 m to obtain a primary emulsion (w/o). Finally, Tween 20® dissolved in hot water (80°C) was slowly added into the emulsion with constant stir- ring (27,000 rpm) at 80°C for 10 m to produce a double emulsion (w/o/w). To decrease the particle size, the emulsion was sonicated using an Bandelin SonoPlus HD 2070 probe type sonicator (Bandelin®, Berlin, Germany) at 70% amplitude level for 2 min to obtain a nanoemulsion (6). FREEZE-DRYING OF SLN DISPERSIONS The nanoemulsions were frozen at -20°C for 5 h and lyophilized in a Alpha 2-4 LSCplus freeze dryer (Martin Christ Gefriertrocknungsanlagen GmbH, Osterode am Harz, Germany) for 48 h. The SLNs were collected for use in differential scanning calorimetry (DSC) analyses (8). PARTICLE SIZE AND ZETA POTENTIAL MEASUREMENTS The mean diameter, polydispersity index (PI), and zeta potential of each sample were obtained using a Malvern Zetasizer Nano ZS (Malvern Instruments, U.K.) at 25°C. Before all measurements, SLNs were diluted with distilled water (10).
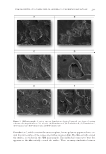
STUDIES OF CAFFEINE-LOADED SOLID LIPID NANOPARTICLES 167 SCANNING ELECTRON MICROSCOPY (SEM) ANALYSIS The nanostructure of the samples was observed using SEM (EVO 40 Carl Zeiss AG, Dresden, Germany). After drying the samples in the oven, they were coated with 15 mm gold-palladium and analyzed using SEM (11). DIFFERENTIAL SCANNING CALORIMETRY (DSC) ANALYSIS DSC (Setaram DSC 131 evo, Caluire, France) analysis was performed to study thermal behavior of the materials. Approximately 6 mg of sample was used for each measurement. The raw materials were analyzed at a speed of 5°C/min in a 20–100°C temperature range. Free caffeine, unloaded (without caffeine) SLNs, and Caf-SLNs were scanned at the same rate speed at a scanning temperature ranging from 20 to 300°C (6). CAFFEINE ENCAPSULATION EFFICIENCY Caf encapsulation effi ciency was measured using an ultrafi ltration method. Caf-SLNs (4 ml) were placed in the upper chamber of an ultracentrifuge tube (Amicon® Ultra-4, 10kDa, Merck Millipore, Darmstadt, Germany). The fi ltered phase containing Caf was freeze-dried and then dissolved in dichloromethane, which was analyzed for Caf amount by using UV spectrophotometry at 273 nm. A calibration curve was generated using six solutions with a concentration range of 10–100 mg/ml, and the correlation coeffi cient was 0.99 [6]. Caf loading effi ciency was calculated using Equation 1 given by: Loading Efficiency (%) = (Actual amount of Caffeine in the SLNs/Theoretical amount of Caffeine in the SLNs) × 100 (1) IN VI TRO RELEASE STUDY Caf release from nanoparticles was performed using the dialysis bag technique (12,13). The dialysis bags (MWCO: 3,5 kDa, Spectrum Laboratories, Inc., CA) were soaked and preconditioned before the experiment. The required amount of formulation (2 ml) was placed into the preconditioned dialysis bag. Then, the dialysis bag was put in 200 ml phosphate buffered saline (pH: 5.5) and incubated in a thermostatic reciprocating shaker maintained at 37 ± 0.5°C and continuously shaken at 100 rpm. An aliquot of 1 ml of release medium was withdrawn at predetermined time intervals (0.08, 0.25, 0.50, 1, 2, 3, 6, 12, and 24 h) and replaced immediately with the same volume of fresh medium to maintain the sink conditions. The concentration of Caf in the aliquot was quantifi ed us- ing high-performance liquid chromatography (HPLC). HPLC ANALYSIS A pharmacopeia method was used for the HPLC analysis of Caf during release studies (14). For this purpose, an Agilent 1260 HPLC system (Agilent Technologies, Waldbronn,
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)