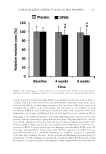
JOURNAL OF COSMETIC SCIENCE 278 Many of these phytochemicals are considered potential contenders to revive tired aging skin and to have wrinkle-combating properties if used topically for suitable duration. However, a search in the scientifi c literature on the Internet yields scanty records on the anti–skin aging properties of date palm kernel extracts (DPKE) in human subjects (2,3). Accordingly, additional studies are necessary to boost the available reports and to estab- lish a considerable body of evidence of anti–skin aging function of DPKE. Assessment of the effi cacy of cosmetic preparations on biophysical characteristics of human skin such as texture, roughness, elasticity, thickness, and wrinkle depth can be achieved by variety of techniques the ideal method should be painless, noninvasive, and provide the specialist with a comprehensive view of skin topography and subsurface structure. Electronic probes with small electronic sensors are now common tools to objectively mea- sure biophysical parameters of skin such as elasticity, hydration, and pigmentation. They can provide clinicians with precise measurements of these parameters in a fast and eco- nomic way (4,5). The advancement in digital technology in recent years enabled noncon- tact measurements of skin relief via optical scanning and digital profi lometric analysis (6). The measuring principle uses scanning the surface with light of different wavelengths and at different angles to collect multispectral optical impressions of the skin surface and using these shape illumination 2D images to reconstruct a stereo 3D image of the skin that can be subjected to photometric stereo analysis by specialized software (7). In this study, we used the Antera 3D® camera (Miravex Limited, Dublin, Ireland) as a means for objective measurement of skin surface parameters (8). The purpose of this piece of re- search, however, was to investigate the antiaging properties of DPKE, formulated in cream form, on the facial skin of 43 healthy volunteers. Assessment of effi cacy was based on measurement of skin biophysical parameters and skin profi lometric analysis by an Antera 3D® multispectral analyzer. S UBJECTS AND METHODS S UBJECTS F orty-three healthy volunteers were enrolled in this study (11 male participants and 32 female participants, age range 39–67 years). Inclusion criteria included the absence of connective tissue or cardiovascular diseases, nonsmokers, and absence of active skin le- sions in the region of interest. Participants were instructed not to apply any cosmetic product or medicinal formulation on the skin area of investigation 1 week before and throughout the study. After fulfi lling the inclusion criteria, each participant was asked to give informed consent by a research coordinator. The study was conducted in accordance with Al Baha University Faculty of Medicine guidelines and the Declaration of Helsinki. P REPARATION OF DPKE CREAM F our hundred grams of date palm kernels (Sukkary variety) were defatted by n-hexane before being ground into a fi ne powder and extracted three times with 800 ml methanol at 4°C for 24 h. After fi ltration and centrifugation, the resultant supernatant was concen- trated under low pressure at 4°C for 3 h to yield the lyophilized material. A cream formula
DATE PALM KERNEL EXTRACT ON FACIAL SKIN WRINKLES 279 containing 5% DPKE was prepared by the method described by Meer et al. (3). In brief, the cream was prepared as an oil-in-water disperse system by heating separately the aque- ous phase and the oil phase containing ABIL® EM90 as a nonionic emulsifi er (Cetyl PEG/ PPG-10/1 Dimethicone, Evonic Industries, Essen, Germany). The methanolic extract of date palm kernels was added in the aqueous phase, and the aqueous phase was added drop by drop to the oil phase at 75°C ± 1.0°C with continued stirring at 2,000 rpm. The emulsion was cooled at room temperature, and the speed of mixer was reduced gradually until the emulsion formed semisolid cream. The same method was used to formulate the base (placebo) cream without adding the DPKE. The composition of the two formula- tions is presented in Table I. The sta bility tests and the physicochemical characteristics of the test and placebo creams were performed at different time points during 8 weeks at 5 ± 0.1°C, 20 ± 0.1°C, 40 ± 0.1°C, 40 ± 0.1°C with 75% relative humidity, and 50 ± 0.1°C (9). PATCH TEST Before beginning the study, a patch test was performed on the skin of 12 volunteers (mean age 46.4 years) to investigate any skin reaction. A 5-cm area was marked on both forearms of each subject, and small amounts of both preparations were applied separately on each forearm then, the region was covered with surgical dressing. Positive skin reac- tions were carefully evaluated after 1, 48, and 72–96 h. The reaction was assessed accord- ing to the International Contact Dermatitis Research Group standard (10). TREATMENT PROTOCO L The region of int erest was the cheek and the temple areas. Both the cream form contain- ing DPKE and the placebo cream were coded and assigned to the right or left side of each volunteer’s face according to a computer-generated randomization list. Participants were asked to apply DPKE and placebo formulations separately on each side of the face twice a day for 8 weeks. Follow-up visits took place every 1 week throughout the study. CLINICAL ASSESSME NT Clinical assessme nt of facial skin was conducted as previously described by using three- point rating scales at baseline and at the end of the study (11). Skin roughness score: 1 = mild, 2 = moderate, and 3 = severe skin texture homogeneity score: 1 = slightly inhomo- geneous, 2 = uneven, and 3 = very uneven skin melanin pigmentation score: 1 = mild, Table I Cream Compositions Phase Placebo cream Cream with DPKE Oil phase Liquid paraffi n (17%) ABIL® EM90 (2.5%) Liquid paraffi n (17%) ABIL® EM90 (2.5%) Aqueous phase Distilled water (q.s 100%) DPKE (5.0%) Distilled water (q.s 100%)
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)