JOURNAL OF COSMETIC SCIENCE 280 2 = moderate, and 3 = severe skin redness score: 1 = mild, 2 = moderate, and 3 = severe. Assessment was performed to study subjects by two independent dermatologists, and repeat assessments were performed to judge the inter- and intrarater reliability, respectively. BIOPHYSICAL MEASU REMENTS Skin hydration, b arrier function, elasticity, pigmentation, and erythema were measured by a multiprobe adapter system (MPA system, Courage and Khazaka electronic GmbH, Koln, Germany). Skin surface hydration was assessed by a Corneometer probe (CM825) (12). The transepidermal water loss (TEWL) was assessed by a Tewameter probe (TM 300), which measures the density gradient of water evaporation from the skin. A micro- processor analyses the values and expresses the evaporation rate in g/m2/h (13). A Cutom- eter probe (MPA 850) measures skin elasticity based on the suction method. The settings of measurements were negative pressure 500 Mbar and suction for 3 s followed by 5 s of release. Both skin fi rmness and elasticity were measured (14). Skin pigmentation and erythema were determined with a Mexameter probe (MX18). Melanin measurement was calculated from the intensity of the absorbed and refl ected light at 660 and 880 nm, respec- tively, whereas erythema was measured at 568 and 660 nm (15). All probes were calibrated daily using standard references, and measurements were taken at baseline, after 4 weeks, and after 8 weeks. Values are displayed as arbitrary units (AU), and each value represents the mean of three measurements obtained from a participant’s same area of skin. SKIN SURFACE DIGI TAL PROFILOMETRY Skin surface profi lometry was performed by an Antera 3D® multispectral analyzer (Miravex Limited) at baseline and after 4 and 8 weeks of treatment. An Antera 3D® cam- era uses multidirectional illumination light to acquire multiple images and reconstructs a three dimensional image of the facial skin by the aid of computer software (8). After a period of 20 min acclimatization in controlled ambient conditions (24°C ± 1°C with 50 ± 10% relative humidity), the camera was placed directly on the external periorbital region over the crow’s feet lines of all subjects and optical scanning images were acquired. After the acquisition of images, several parameters related to skin topography were measured, including wrinkle depth, indentation index of fi ne lines (marginal size less than 1.5 mm), folds (marginal size less than 2.5 mm), and wrinkles (marginal size less than 5.0 mm). In addition, texture roughness, skin pigmentation (melanin), and redness (hemoglobin) were also assessed. The identical area was digitally identifi ed at each session, and values are presented as the result of triplicate measurements from the same area. STATISTICAL ANALYSIS Th e statistical analysi s was performed using SPSS version 15.0 (SPSS Inc., Chicago, IL) Data were represented as the mean ± SD. Multiple comparisons between means were performed as appropriate by using the paired t-test or the one-way analysis of variance (ANOVA) followed by Tukey post hoc test. In all the tests, a p-value of less than 0.05 was considered signifi cant.
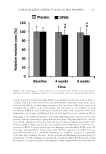
DATE PALM KERNEL EXTRACT ON FACIAL SKIN WRINKLES 281 RESULTS Only 36 subjects of 43 p articipants completed the study (seven male participants, age range 41–55 years, and 29 female participants, age range 40–65 years). Reasons for drop- ping out from follow-up visits were given as holiday time, tiredness, and other family issues rather than any problem with the cream preparations. A preliminary patch test revealed no skin reactions observed at 1, 24, and 96 h after removing the test materials in all 12 subjects. The physicochemical characteristics of both the placebo and test creams, kept at different temperatures for 8 weeks, are shown in Table II. The color was pearly white with no changes in homogeneity or phase separation up to 50 ± 0.1°C and up to 40 ± 0.1°C with 75% relative humidity. The presence of the lipophilic emulsifi er ABIL® EM 90 stabilized the emulsions at high temperatures. CLINICAL ASSESSMENT Clinical assessment of facial skin appearance was conducted at baseline and after comple- tion of the study (i.e., after 8 weeks). Based on a three-point scale evaluation, there were statistically signifi cant improvements in skin roughness, texture homogeneity, and melanin pigmentation. Skin redness levels were not signifi cantly altered in all subjects (Table III). BIOPHYSICAL MEASUREMENTS Meas urements of objective ski n parameters with a multiprobe instrument over 8 weeks revealed progressive increases of surface hydration and elasticity together with a progres- sive decline in both dermal pigmentation and TEWL in DPKE-treated skin compared with placebo cream. Figure 1A illustrates changes of skin hydration at three time points of study. At baseline, the mean values of skin hydration were 41.21 ± 10.32 and 42.10 ± 9.64 AU of placebo and DPKE creams, respectively. After 4 weeks, these values rose to 43.71 ± 11.62 and 52.92 ± 10.25 AU, respectively, and after 8 weeks, the values were 44.21 ± 10.42 and 52.23 ± 11.54, respectively (p 0.01 vs. placebo and p 0.001 vs. baseline for the two time points). Figure 1B illustrates changes of TEWL at three time points of the study. At baseline, the mean readings of TEWL were 12.23 ± 3.11 and 11.76 ± 2.92 g/m2/h of placebo and DPKE creams, respectively. After 4 weeks, these values were 11.72 ± 2.52 and 10.43 ± 2.41 g/m2/h, respectively, and after 8 weeks, the Table II Physicochemical Characteristics of Placebo and DPKE Creams Kept at Different Temperatures for 8 Weeks Color Texture Homogeneity Phase separation Immediate skin feel 5–40 ± 0.1°C White Smooth Homogeneous No Refreshing, cool, no grittiness or greasiness 40 ± 0.1°C with 75% relative humidity White Smooth Homogeneous No Refreshing, no grittiness or greasiness 50 ± 0.1°C White Smooth Slight liquefaction Slight Refreshing, no grittiness or greasiness
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)