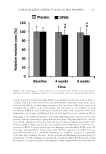
JOURNAL OF COSMETIC SCIENCE 314 One of the most popular and valuable natural products is coffee. Because of the high consumption of coffee as a beverage, the coffee industry is categorized as one of the largest industries in the world (6). Besides its good taste, coffee is benefi cial for human health because many studies have shown that coffee contains phenolic compounds such as chlorogenic acids, which show antioxidant activity (7,8). The coffee industries also produce a large number of by-products, such as coffee spent grounds and coffee silver- skin (9). Coffee silverskin is produced as a by-product during the roasting process of coffee beans. It is a thin layer surrounding the coffee bean, and it is usually still at- tached to the coffee bean even after the depulping and dehulling process of the coffee cherries. When the coffee beans are heated at high temperature during the roasting process, the coffee beans will crack because of loss of moisture content, resulting in the detachment of the coffee silverskin from the beans. Because of its lightness, the coffee silverskin escapes from the roasting container through the roaster exhaust or cyclone, and is usually disposed of as waste. However, recent studies on the revalorization of coffee silverskin reported that coffee silverskin contains phenolic compounds and shows high antioxidant activity (10–12). Bresciani et al. (13) reported that chlorogenic acids are the sole phenolic compounds in coffee silverskin. The studies on the extrac- tion of coffee silverskin mostly used a mixture of Robusta and Arabica coffee silverskin because the roasting industry usually blends these two types of coffee. However, our recent study showed that Robusta coffee silverskin showed a higher phenolic content and, thus, a higher antioxidant activity than Arabica coffee silverskin (14). This re- sult is in accordance with the study conducted by Farah et al. (8) who reported that Robusta coffee beans contain a higher amount of chlorogenic acid than Arabica coffee beans. Rodrigues et al. (15) reported that coffee silverskin was an effective ingredient in the improvement of skin hydration and fi rmness. De Hond et al. (16) studied the use of coffee silverskin extract to protect accelerated skin aging. In spite of the high antioxidant activ- ity of coffee silverskin and its great potential for industrial application, to the best of our knowledge, there is no study on the development of skin care products containing coffee silverskin extract as an antioxidant. Moreover, a proper extraction method and the opti- mization of the extraction condition to obtain an extract of coffee silverskin with a high phenolic content and a high antioxidant activity are not studied yet. Furthermore, the extraction of coffee silverskin was usually conducted by maceration process, which means the raw material is immersed in a solvent without any mechanical agitation. This conven- tional method takes a long time to extract the phenolic compounds, and results in a low phenolic content. In t his work, coffee silverskin of Robusta type was extracted using a hydroalcoholic solvent with the aid of mechanical agitation at various extraction times and tempera- tures with the purpose to maximize the phenolic content and the antioxidant activity of the coffee silverskin extract. The extract solution was then dried using a spray dryer to produce coffee silverskin extract in powder form. The study on the effect of drying of the extract is important because extracts are usually produced in powder form. Furthermore, to develop an antioxidant skin care product, a basic skin gel was prepared and the coffee silverskin extract powder was added as an active ingredient in the skin gel. The total phenolic content and the antioxidant activity of the skin gel were measured to study the effect of the addition of the coffee silverskin extract to the skin gel.
DEVELOPMENT OF ANTIOXIDANT SKIN GEL USING COFFEE SILVERSKIN 315 MATE RIALS AND METHODS MATE RIALS Robu sta coffee silverskin was obtained from a coffee roasting company located in Bandar Lampung, Indonesia. Technical grade ethanol (96% v/v) was purchased from PT Sumber Abadi (Tangerang, Indonesia). Sodium carbonate (BDH, London, England), Folin– Ciocalteu reagent (Merck, Darmstadt, Germany), gallic acid (GA) powder (Aktin Chemical, Chengdu, China), aluminum chloride (Merck), potassium acetate (BDH), 2,2-diphenyl- 1-picrylhydrazyl (DPPH, Sigma-Aldrich, St. Louis, MO), and analytical grade ethanol (Smart Lab, Bogor, Indonesia) were used. Chemicals to prepare the basic skin gel were carbomer 940, propylene glycol, methylparaben, and propyl paraben, all purchased from PT Intralab Ekatama (Bogor, Indonesia), whereas triethanolamine (TEA) and ethylene diamine tetra acetic acid (EDTA) were purchased from PT Sumber Abadi. Distilled water was used. EQUI PMENT Stan dard laboratory equipment was used in this work, namely, hot plate (Cimarec, Waltham, MA), beaker glasses, Erlenmeyer fl asks, magnetic stirrer, thermometer, volumetric glass (Pyrex, Corning, NY), water bath shaker (Memmert, Schwabach, Germany), micropipette (Eppendorf, Hamburg, Germany), micropipette tips, cuvette (Brand GMBH, Wertheim, Germany), rotary vacuum evaporator (IKA HB 10, Shanghai, China), spray dryer (BUCHI mini spray dryer B-290, Flawil, Switzerland), analytical balance (Ohaus PA214, Parsip- pany, NJ), UV-Vis spectrophotometer (Genesys 10-S, Waltham, MA), vortex (Vortex- Genie 2, St. Louis, MO), fi lter instrument, Whatmann fi lter paper 1001 125 (GE, Little Chalfont, Buckinghamshire, UK), desiccator, moisture content analyzer (Sartorius MA35, Goettingen, Germany), mixer (IKA Labortechnik, Staufen, Germany), pH meter (Lutron pH-208, Taipei, Taiwan), and viscometer (Brookfi eld DV-E, Lorch, Germany). EXTR ACTION PROCEDURE Firs t, the coffee silverskin sample was rinsed with water and dried in an oven at 40°C. The dried sample was milled using a mixer grinder to reduce the size. Then, 100 mL of water– ethanol mixture with a weight ratio of 50:50 was poured into a glass beaker and heated using a hot plate. The temperature was varied at 30, 40, 50, and 60°C. When the desired tem- perature of the solvent was reached, 2 g of coffee silverskin were poured into the solvent, and the extraction process was carried out by stirring using a magnetic stirrer at a speed of 350 rpm. Thus, the weight ratio of the coffee silverskin and water–ethanol solvent was 1:50. The beaker was covered with aluminum foil to prevent loss of solvent because of evapora- tion during heating. The extraction time was varied at 5, 10, 20, 30, 40, and 60 min. The extract solution was then fi ltered using a fi lter paper and stored in a refrigerator before analysis. ANALYS IS OF TOTAL PHENOLIC CONTENT AND ANTIOXIDANT ACTIVITY The to tal phenolic content was determined by using the Folin–Ciocalteu method. First, the F olin–Ciocalteu reagent solution was made by diluting concentrated Folin–Ciocalteu
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)