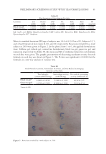
73 ENCAPSULATED TTO IN FACIAL CREAMS MATERIALS AND METHODS MATERIALS Aqueous 60% β-cyclodextrin solution (hydropropyl-beta-cyclodextrin, HPbCD, I.R.A. Istituto Ricerche Applicate, Italy) and TTO (Main Camp Natural Extracts Pty Ltd, Australia), commercially labeled as “PHYTODERMINA LIFTING” and “Tea Tree Oil (M alternifolia)” respectively, were supplied by Cellco Chemicals SA. ELECTROHYDRODYNAMIC PROCESS (ELECTROSPINNING) The electrohydrodynamic process was performed in the electrospinning apparatus FluidNatek®, equipped with a variable high-voltage 0–30 kV power supply (Bioinicia S.L., Valencia, Spain). Cyclodextrin solution (matrix) and TTO (encapsulated material) were electrospun in a horizontal coaxial electrospinning setup. Cyclodextrin and TTO were placed in a different plastic syringe (10 mL, 14.22 mm diameter) each, with horizontal orientation on a pump that digitally control them, with their needles vertically directed toward the collector. The electrode emitting positive polarity of the high-voltage power supply were connected to the needles and an aluminum foil sheet was attached to a copper grid, placed 15 cm from the capillary tip, used to collect the electrospun fibers. The electrospinning experiments were carried out at room temperature in air and the flow rate of cyclodextrin solution was set to 0.5 mL/h and of TTO to 0.4 mL/h, while the voltage was set to 26.1 kV. In optimum conditions the encapsulation efficiency was 75% and the amount of TTO was extracted from weighted nanofibers (collected after 2 hours) using ethanol as solvent. The encapsulation efficiency was then determined using a UV– spectrophotometer at 225 nm. SCANNING ELECTRON MISCROSOCPY Morphology of TTO encapsulated microfibers was evaluated using scanning electron microscopy (SEM) (Quanta 200, FEI Oregon, USA, voltage 12.5 kV, LFD detector, Spot size 4.5, magnification ×12000). Prior to SEM, a 13.23 nm coat of gold layering was applied to make the surface reflect the electron beam (SC7620 Mini Sputter Coater, Quorum Technologies, West Sussex, UK /105 s, 18 mA, 1KV). PREPARATION OF FACIAL CREAM SAMPLES The facial cream was based on a natural olive-oil–derived emulsifier and a natural wax containing 12% natural and synthetic oils stabilized by natural polymers (carrageenan, cellulose gum, xanthan gum) (sample C). Sample C did not contain TTO. For the formulation of the samples containing encapsulated TTO (sample T) the required quantity of powdered encapsulated fibers were added in the basic cream to achieve 0.5% TTO quantity in the final formulation. The percentage of TTO in the final encapsulated fibers was approximately 37%.
74 JOURNAL OF COSMETIC SCIENCE STORAGE CONDITIONS Immediately after their formulation (time zero, control samples), facial creams were sampled in the laboratory and stored in glass containers in temperature-controlled cabinets at three selected storage temperatures: 5°C, 25°C, and 45°C and analyzed to verify their stability over storage time. A storage temperature of 45°C was selected to facilitate an accelerated shelf-life testing protocol and all samples were stored for 50 d in total. For all tests cream sampling was performed after their formulation (time zero, control samples) and after 30 and 50 d of storage in all the different temperatures. MICROBIOLOGICAL ANALYSIS For microbiological enumeration, a representative sample of 1 g of each sample was aseptically taken from each product and placed in 9 mL sterilized Ringer solution (Ringer tablet, Merck code 1.15525, Darmstadt, Germany), thus giving a 10:1 dilution. Samples were homogenized using with a vortex at room temperature. The outside surfaces of all containers were swabbed with ethanol (70% v/v) before opening. Sampling of the creams was carried after 30 and 50 d of storage. The microbiological stability of all products was examined. Total count and yeasts/molds (YM) were determined, as well as the pathogens, S aureus, P aeruginosa, E coli, and Salmonella spp. Compact dry plates, a ready-to-use test method, was selected for the microbiological analysis. For all plates, typical colony types and morphology characteristics associated with each growth medium were examined. The number of viable germs in a culture can be ascertained by determining the number of colonies forming units (cfu/g), with the colony counting technique. Plate reading and colony count were reported in cfu/g (34). TOTAL BACTERIAL COUNT Compact Dry TC is a medium contains nutrient standard agar, suitable for total viable bacterial count. Aerobic bacterial colony counts were made by spreading 1 mL of each 10:1 diluted sample on Compact Dry TC medium for total count (Compact Dry TC, Hyserve GmbH &Co. KG, Uffing, Germany) and incubated at 37°C for 48–72 hours. Each assay was performed in duplicate. After incubation, the number of colonies was recorded for each plate. In samples with low counts, the count was recorded as 100 cfu/g of product. YEASTS/MOLDS YM are both fungal spp. and many, as with other microorganisms, can cause serious infections of the immunocompromised. YM can be differentiated by color development. A ready-to-use plate labeled Compact Dry YM contains a medium with a chromogenic enzyme substrate (XPhos) that changes to blue when many of the yeasts are detected. During the 3-dimension growth of yeast and mold, fluffy colonies with characteristic colors are formed while antibiotics inhibit the growth of bacteria. For the enumeration of YM 1 mL of each 10:1 diluted sample was spread on Compact Dry YM ready-to-use plates for yeast and mold (Compact Dry YM, Hyserve GmbH &Co. KG, Uffing, Germany) after
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)