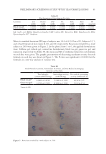
77 ENCAPSULATED TTO IN FACIAL CREAMS An important contamination of the cosmetics during usage refers to Staphylococcus spp. mainly due to the contact with users’ hands as presented by Campana et al. (34). The recovery of these potential pathogenic spp. suggested that the preservative system was not effective, in contrast to the samples of the present study, where it is apparent that they were not contaminated during storage, due to proper handling and storage conditions. Behravan et al. reported the need for a system to prevent microbial growth (38). They investigated the presence of microorganisms in a variety of sealed and used cosmetic creams and found that the contamination with Gram-positive bacilli, S aureus, was much greater in all samples, while some Gram-negative E coli were also isolated from both the used and unused creams. They were found to be higher for used cosmetic creams (45%, 38%, and 8%, respectively) than for unused creams (38%, 25%, and 0%, respectively). In addition, 17% of the unused creams were found to not be viable microorganisms, while only 10% of the creams used did not contain viable microorganisms. Most of the studies did not report on Salmonella spp. infection due to use of contaminated cosmetic preparations, which was the case in our study. Specifically, this could be attributed to several compounds (especially plant origin) that are commonly used in preparation of cosmetics and prevent Salmonella spp. growth (40). Table III Counts of Bacteria Associated With Cosmetic Creams Stored at 45°C for 50 d Sample Total bacterial count YM P aeruginosa S aureus E coli Salmonella C 2 log cfu/g 1 log cfu/g absence/g absence/g absence/g absence/g T 2 log cfu/g 1 log cfu/g absence/g absence/g absence/g absence/g Figure 1. SEM of encapsulated TTO microfibers.
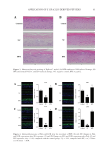
78 JOURNAL OF COSMETIC SCIENCE Microbial analysis of the examined C and T cream samples revealed a bioburden 10 cfu/ mL. Such data were also obtained by Oliveira et al., after performing similar microbiological tests to different cosmetic formulations (31). After performing the essays for P aeruginosa, S aureus, E coli, and Salmonella spp. no growth of these pathogens was observed. Thus, we conclude that despite the occurrence of some contamination during the manipulation of the cosmetic creams, the microorganisms did not survive because of a proper composition on the formulations, ensuring their quality. The results obtained for all samples prove them to be safe for use regarding their microbiological aspects (31). SEM analysis (Figure 1) showed a homogeneous grid of microfibers with an average diameter of 0.2 μm. Accelerated testing performances and kinetics of pH profiles can be considered the most important aspects of chemical stability in pharmaceuticals (41). Moreover, regarding emulsions and especially cosmetic creams, pH is a crucial parameter that affect the effectiveness of the formulations. The skin surface has a slightly acidic pH, between 4 and 6 with reported average value of around 5. Lambers et al. describe that skin surface pH evolves to a “natural” average value of 4.7 (32). Thus, cosmetic creams should have a pH close to these values. The resulted pH values of the examined samples stored at 5°C, 25°C, and 45°C were in the reported values range between 4.54 and 4.86, as shown in Figure 2. More specifically, at t =0 for samples C and T the pH values were 4.86 and 4.72, respectively. The storage time and temperature do not significantly affect these values, as for sample C the values after 50 d slightly decreased to 4.75, 4.64, and 4.54 for samples kept at 5°C, 25°C, 45°C respectively. While the respective values for sample T were 4.75, 4.72, and 4.56 for samples kept at 5°C, 25°C, 45°C respectively. Regarding the incorporation of TTO, the pH measurements indicate minor differences comparing to C samples. Thus, as a function of time and storage temperatures, pH values for all the examined samples indicated that the examined cream samples were stable. Variation in total color change (ΔΕ,) lightness (L*), redness (a*), and yellowness (b*) values with storage for the samples C and T stored at 5°C, 25°C, and 45°C are shown in Table IV– VI. Results of the color measurement showed that the L* present similar values between C and T samples for all the storage temperatures and time, whereas the highest values of a* and b* were noted for creams C, although the differences between C and T samples were not significant regarding a* and b* values. It can also be observed that with storage time, L* and b* values remain more or less stable, with negligible decrement, for samples in all stored temperatures. The highest decrement for both a* and b* values was observed for T samples after storage in 25°C. In all cases, L* values were between 65 and 67.16, indicating the emulsions were whitish (i.e., bright) in color. In terms of hue, the samples at all storage temperatures present negative a* values ranging from –1.26 to –1.67 that indicate green color. The b* values were all positive, ranging from 4.56 to 5.39, indicating samples had a yellow hue. The similar values of samples containing TTO (T) and the base cream (C) are indicative of the successful encapsulation procedure of TTO that prevent its slight yellow color to alter the color of the final formulations. Tables IV–VI and Figure 3 show how ΔΕ presents an increasing general trend during storage time. The observed differences between samples C and T are due to the addition of TTO, however these differences are not significant. Finally, the observed differences in color between samples T and C were negligible, indicating the stability of formulations and the controlled release of TTO during cream storage.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)