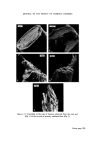
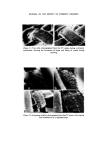
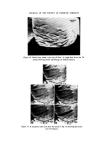
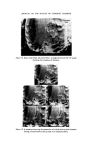
286 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS the concentration of contaminants has reached a level to which the chosen method of analysis is sensitive. In the case of total viable counts using the pour plate technique this means that a product containing less than, say, 30 organisms g-X would be unlikely to be found to be contaminated. This has important implications when selecting the time after manufacture at which a product should be sampled for contaminants. For example, the experi- ments reported here show that in the model shampoo system E. cloacae had a generation time (the time required for the bacterial population to double in size) of between 70 and 92 min. Now in a manufacturing unit where batches of 2000 kg of shampoo are made there would need to be a total of 6.0 x 107 bacteria present for contamination to be detectable in the bulk, i.e. 30 bacteria per gram. Contamination would not be detectable until at least 30 h after manufacture if the original inoculum was 50 bacteria and the generation time 90 min. Even with an initial inoculum of 5 x 105 bacteria, more than 10 h would elapse before detectable numbers of organisms would be present. Thus except in those cases where gross contamination of a product has occurred microbiological examination of either the bulk product or the product in the final container immediately after manufacture is unlikely to reveal the presence of bacteria. Thus products should be examined not only on the day of manufacture but also some days later 7 days would be a convenient period. In view of our observation that in a model shampoo with a low inoculum bacteria become detectable within 24 h, an incubation period of 7 days might seem excessively long. However, in some products the phase of rapid multiplication, the logarithmic phase, may be preceded by a lag phase in which no multiplication occurs and also the generation time may, under less favourable conditions, be considerably longer than 90 min. Since it may take a further 7 days for the results of microbiological examination to be known it may be thought necessary in the future to pre- vent distribution of finished products until 2 weeks after manufacture. How- ever, this will depend upon experience, the ease or otherwise of recalling products once they have left the factory and the risk the manufacturer is prepared to take. The ability of small numbers of bacteria to contaminate large volumes of shampoo helps to explain the occasional observation that contamination may be found in some packs from a batch but not others. If the original inoculum is very small or unevenly dispersed then only a certain proportion of all packs will contain bacteria capable of multiplying and giving rise to detectable numbers of contaminants. The length of the logarithmic phase, i.e. the time required for contaminants in a product to achieve their maximum
WATER-BORNE BACTERIA AND SHAMPOO SPOILAGE 287 number, will increase with an increase in batch size. However, during this phase the longer a product is stored prior to packing the greater will be its bacterial population and the greater the proportion of packs containing contaminants. So products should be packed as soon as possible to reduce the frequency of pack contamination. If storage prior to packaging is un- avoidable, viable counts should be performed to establish the advisability of packaging. The inability of total viable counting techniques to detect less than about 30 bacteria per gram of product might be considered to be an argument for their replacement by other more sensitive techniques, e.g. inoculation of a liquid nutrient medium with a sample of product or filtration methods. How- ever, unless the complete absence of contaminants or the absence of specific types of organisms is required these methods will be of doubtful value since a manufacturer will not reject a batch of product, until it has been shown that the contaminants present are capable of multiplying to an unacceptable level. Growth in liquid media has other disadvantages: chance contamina- tion may give rise to false positive results, no information concerning the numbers of bacteria in the product is provided, bacteria capable of growth in the product may be outgrown by those of no significance from a product contamination viewpoint and a standard of sterility is imposed on the product. Experimental inoculation of shampoos with bacteria derived from mains- water indicates that the bacteria capable of growth represent only a small proportion of the total population carried by water (our experience is of course with water from a single source). Using Probability Tables (26) by extrapolation, our results suggest that the number of bacteria capable of multiplying in our model shampoo system is approximately 5 in 100 litres (less than 0.000055/0 of the total number of bacteria present). However, a 2000 kg batch of our model shampoo would contain 1228 1 of water or 60 bacteria capable of surviving and multiplying in this product. This is, according to our results, a sufficient number to contaminate it. Had we used more inhibitory detergent systems (e.g. monoethanolamine lauryl sulphate) or had we used test bacteria less well adapted to growth in sodium lauryl ether sulphate, it is probable that we would have found that more than 50 bacteria were necessary to contaminate our model shampoo. Similarly the frequency of occurrence in water of bacteria capable of sur- vival and multiplication in a product will be least when that product provides an inhospitable environment for bacteria. But when selecting water treat- ment devices or designing plant cleaning and disinfection procedures it is
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)