MICROBIOLOGICAL QUALITY CONTROL 317 and in sensitive product areas were checked by R & D personnel. Only then was final testing of finished packed products introduced, product by product, with priority given to products known to be at risk in the factory (aqueous 'cold mix' products) and if contaminated those of direct risk to the con- sumer, for example, baby toiletries and oral non-sterile pharmaceuticals. The final check was by means of a total viable count per ml or gram of product, with the count carried out in such a way as to ensure the viability of as wide a range of organisms as possible and in the arithmetical treatment of numbers an inbuilt bias for counting high. In the early days of the programme no attempt was made by QC personnel to identify organisms, their role was purely that of quantification. If high counts were met, R & D personnel would visit the plant and after investigation pinpoint the source of contamination. In this way everyone, QC, Production and R & D learned from errors and mistakes. A plant with- out problems teaches nothing but merely adds to a sense of false security. The author has previously outlined the basic techniques for total viable count (1), but several points bear mention yet again. The aim of a viable count is to provide a series of conditions under which each viable microbial unit in a material will give rise to a discrete visible colony which can then be counted. It is easier in microbiology to determine sterility (under defined conditions) than it is to provide accurate assessment of counts between sterility and 100 organisms per g or ml. This is due to the fact that very few products could be plated directly without dilution and such dilution rapidly affected the reliability of the count. For example, with accept/reject limits at 100 a dilution of 1 in 10 will require a decision on counts of less than 10. This is an order of magnitude lower than the density usually accepted as optimal. It was fortunate that right at the beginning two temperatures of incuba- tion (24øC and 37øC) were used rather than one as in the US Pharmaco- poeia (2) (32øC). Bacteria such as Achromobacter which do not grow above 30øC have been a major problem over the years. Likewise, it was ensured that organisms with different growth requirements were catered for by using two non-specific growth media, nutrient agar for bacteria and Sabouraud dex- trose agar for fungi. With various products at one time or another, spread plates, pour plates, membrane filtration and most probable number methods have been used for viable count. Where a product produced in many centres has differing standards imposed by various governments, as a matter of good quality assurance the highest standard required has been imposed as the minimum company
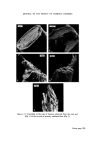
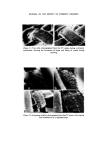
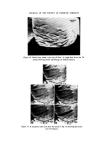
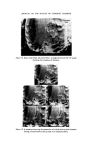
318 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS standard for that particular product. This has ensured greater flexibility in supplying different countries from one centre. As a result of 4 years' experience there is good confidence that because of the MQC programme the products leaving the plants are pure and whole- some. ORGANISMS WHICH HAVE CAUSED PROBLEMS A review of the records places the culprits in the following order of importance. 1. Achromobacter 2. Pseudomonas. 3. Bacillus. It is surprising perhaps that the Enterobacteriaceae and Micrococcaceae do not figure in our list but the survival value of pathogens in plants such as Escherichia coli and Staphylococcus aureus is essentially nil and they have never been a cause of control breakdown. Without doubt in places as far apart as Liverpool, Madrid and Johannesburg the Achromobacter have been the major problem. Both Achromobacter liquefaciens and A. guttatus have been isolated from mains water, deionized water, filling lines and finished product. They have caused a major rethink in the provision of deionized water to points in the plant and the use of filtered and boiled water for rinsing procedures. Pseudomonas aeruginosa and another Pseudomonas spp. have been responsible on two occasions for the phenomenon of adaption. On these two occasions the organisms were metabolizing a detergent present in the two products and were totally resistant to the paraben pre- servation in one case and benzalkonium chloride in the other. One plant had to be closed for sterilization and in the other the filling equipment was scrapped and replaced. This latter example serves to illustrate the problems that can be met in the factory environment when Pseudomonas aeruginosa is present. J Early in 1972 an overseas plant reported contamination in an aqueous topical non-sterile pharmaceutical. Filled stock was contaminated with an organism growing at both 24øC and 37øC on Nutrient Agar. This organism was present between 105 and 10 ? ml -• and produced an intense green pigment. It was rapidly established that bulk product and packaging material were both free from contamination and it was, therefore, the filling equipment
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)