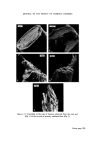
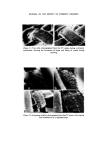
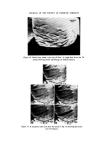
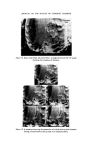
MICROBIOLOGICAL QUALITY CONTROL 319 which was the most likely source of contamination. An immediate monitor- ing of the filling equipment, raw materials and water, personnel, packaging and bulk product was undertaken. Contamination was only found 'down- stream' of a rotation pump feeding bulk into the stationary line. Flushing through the system with sterile water and counting at the filling nozzles showed that approximately 10 a organisms ml -x were being delivered. The entire filling equipment was dismantled and replaced with stainless steel parts throughout and the old pump was scrapped. A daily disinfection procedure was drawn up and process workers instructed in its use, With this new equipment and standard operating procedure the plant has been free of all contamination for over 2 years. The analysis of plant water showed that contamination was minimal and did not contain Ps aeruginosa but there were Ps spp. present. The Ps aeruginosa isolated produced intense green pigmentation on nutrient agar, the pigmented colonies showed fluorescence and the plates smelled of methylamine. Oxydase was positive, it grew on and in Centrimide agar and broth and was typed as Pyocine 36 B. When inoculated from contaminated product it could utilize Triton WR1339 as its sole source of carbon. Triton WR1339 is the non-ionic detergent present in the topical product. Prompt response to the initial finding, the quarantining of stock and immediate clean up procedures prevented a major production disaster. Final rinsing procedures after disinfection but before filling are now carried out with boiled deionized water or deionized water filtered through a final bacterial filter at the point of use. The use of mains water or non- sterilized deionized plant water does allow inoculation of equipment with various waterborne bacteria. The level of inoculation is extremely low but where there has been a breakdown in routine production the frequency of contamination problems rises considerably. For example a cosmetic lotion had two bulk batches pooled to adjust viscosity and on filling was shown to be contaminated with Achromobacter liquefaciens which was present in the plant water. The Achrornobacter has been found somewhat more resistant to the usual parben preservatives so now this organism is included routinely in challenge tests carried out by R & D. Achrotnobacter is defined as Gram negative motile or non-motile rods which do not form pigment and whose optimum growth temperature lies at 30øC or below. An exact name has only been put to two species but the vast majority of isolates are of these two species, A. liquefaciens and A. guttatus. A. liquefaciens liquifies gelatine, A. guttatus does not, and neither produce nitrites from nitrates.
320 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS The Bacillus group of organisms have been infrequent contaminants of products and when the occurrence has been investigated no reasonable explanation has been made of that occurrence. These organisms are only remarkable for the low level of contamination found but because of the contamination being in the spore phase this low level of contamination remains in the product due to the preservatives present having no sporocidal activity but merely preventing exsporulation and growth. All species isolated so far have been aerobic in spite of considerable expenditure of time in attempting the isolation of Clostridia from talcs and similar materials. Other than Ps aeruginosa classical pathogens such as E. coli, Salmonella spp. or Staphylococcus aureus have not been isolated. When required by regulation to certify products as being free from these organisms but in the absence of approved test methods the following test structures were utilized. E. coli An aliquot is taken from the product sample and is used to inoculate a primary enrichment broth (Digest or Glucose) which is incubated at 37øC for 2 days. This broth is then subcultured onto a selective media containing bile salt, lactose and indicator (MacConkey Agar) at 37øC for 24 h. Any Gram negative lactose fermenting colonies are inoculated into an equivalent broth (MacConkey Broth) and incubated at 44øC for 24 h. The production of both acid and gas at this temperature is considered to be presumptive of E. coli. Ps aeruginosa An aliquot is taken from the product sample and is used to inoculate a primary selective/inhibitory broth (Digest with 0.02•o Cetrimide) which is incubated at 37øC for 2 days. This broth is subcultured onto an equivalent agar (Nutrient with 0.03•o Cetrimide) at 37øC for 24 h. Any Gram negative colonies growing on this plate which smell of trimethylamine and display greenish pigment and/or fluorescence under a UV lamp are considered to be Ps aeruginosa. Salmonella spp. An aliquot is taken from the product sample and is used to inoculate a primary enrichment broth (Digest or Glucose) which is incubated at 37øC for 2 days. This broth is subcultured into a selective/inhibitory broth (Selenite or Tetrathionate) at 37øC for 24 h. This broth is then subcultured
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)