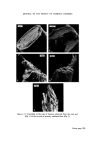
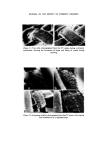
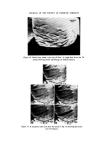
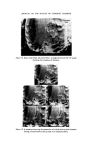
MICROBIOLOGICAL QUALITY CONTROL 321 onto an inhibitory/differential agar (Wilson and Blair bismuth sulphite agar) at 37øC for 48 h. Any Gram negative colonies growing on this plate which are black or green, have a metallic sheen or a diffuse black sulphide deposit around the colony are presumed to be Salmonella spp. The most certain and rapid confirmatory test is to subculture suspect colonies onto Nutrient agar and test the colonies after 24 h growth with appropriate polyvalent antisera. Staphylococcus aureus An aliquot is taken from the product sample and is used to inoculate a primary enrichment broth (Digest or Glucose) which is incubated at 37øC for 2 days. This broth is subcultured onto an inhibitory/differential agar (Tellurite-egg or Vogel-Johnson) at 37øC for 48 h. Any Gram positive colonies growing on this plate (Vogel-Johnson) which are small, black surrounded by yellow zones are presumed to be Staph. aureus. For confirmation, subculture suspect colonies into tubes of mamma- lian plasma (0.5 ml) and incubate at 37øC. Examine at 3 and 24 h. Coagula- tion constitutes a positive test. LESSONS LEARNED FROM THE MICROBIOLOGICAL QUALITY CONTROL PROGRAMME It has been recognized that only sterile products are expected to be free from detectable organisms and it is, therefore, important to recognize that non-sterile products can and do have organisms present in any product. It is, therefore, necessary to draw up techniques of viable counting with full laboratory instructions to determine such contamination and to set limits on that contamination. The microbiological quality of any raw material that is used in the manu- facture of products should be known and determined both in the develop- ment laboratory and in the factory. It is important to draw up realistic specifications for raw materials and great assistance may be had from most suppliers in doing this. It is obvious that during the manufacture of a non-sterile product, the process should be so designed, so that the microbial content does not increase. The function of a preservative is to prevent proliferation of an accidental contamination, not to free production from rigid hygiene standards and
322 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS because of the importance of good preservation consideration is now being given to the introduction of QC tests to determine the components of the preservative system into the final product specifications. Packaging components have been found to play an important role in the microbiological integrity of a product and in the past insufficient attention was shown to the microbiological aspect when packaging was chosen. It is necessary to stress that not only could the components of the packaging introduce undesirable organisms into formulations but also they can interact chemically and physically with formulation ingredients (including pre- servatives) and thus change the susceptibility of the final formulation to bacterial insult. The package is part of the product and must be present for proper formulation evaluations. Finally, without doubt, the provision of pure wholesome plant water is the number one priority of a microbiological control programme. The majority of incidents can be traced to water as the culprit. (Received: 30th May 1974) REFERENCES (1) Breach, G. D. Microbiological quality control. Paper 9 Symposium Evaluation of Product Performance (Society of Cosmetic Chemists, Nottingham, November 1973). (2) The Pharmocopeia of the United States of America, 18th Revised (1970) (Mack, Easton PA 18042).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)