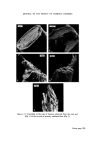
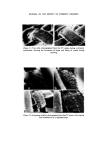
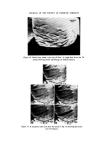
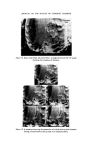
J. Soc. Cosmet. Chem. 26 315-322 (•975) ¸ 1975 Society of Cosmetic Chemists of Great Britain Microbiological a case history quality control G. D. BREACH* Presented on 28th August 1974 in London at the IFSCC VIIIth International Congress on 'Cosmetics--Quality and Safety' organized by the Society of Cosmetic Chemists of Great Britain Synopsis--A history of the introduction of MICROBIOLOGICAL QUALITY CONTROL (MQC) into factories manufacturing non-sterile pharmaceuticals, TOILETRIES and COS- METICS throughout Europe and Africa is outlined. The development of a microbiology manual and appropriate standard operating procedures to up-grade GOOD MANUFACTUR- ING PRACTICE (GMP) is explained. Problems with particular products and organisms are discussed, especially the presence of Achromobacter and Pseudomonas organisms in plant water in some sites. The inter-relationship of QC, R & D, and Production which enabled MQC to proceed smoothly is emphasized as is the application of one overall 'in house' standard. THE INTRODUCTION OF MICROBIOLOGICAL QUALITY CONTROL We viewed microbiological control as a much needed extension of a quality assurance programme and the techniques, methods and standards which were developed were designed to control and improve hygiene standards in the plant. Where official government standards have been intro- duced these have been viewed as additive, rather than as a replacement to the 'in house' standards. For example, a government regulation which requires the absence of a particular species of bacteria from a product, utilizes a specific test designed for that organism and therefore it is felt that * Vick International, 250 Bath Road, Slough, Bucks. 315
316 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS the product's ability to pass such a test does not measure the overall hygienic production of the product or indeed the actual level of bacterial contamina- tion present. It was also felt to be necessary to incorporate into the stability programme regular microbiological analysis, so that knowledge could be gained of the microbiological life-span of products and their preservative systems and sufficient data acquired for meaningful co-operation with government bodies, dealing in practicalities rather than airy-fairy theories. All the QC managers were involved from the outset of the programme so that due economy in materials and time could be obtained. At the outset it was felt that it should be possible to introduce MQC to non-microbio- logically qualified staff if methods and the results obtained by those methods did not contain elements of judgment and opinion. By this it was meant that the staff and premises available at that time were sufficient to initiate and maintain a programme of microbiological testing without: (a) employing microbiologists in each plant (b) installing a separate microbiological laboratory in each plant. To this end a manual was prepared by qualified microbiologists in R & D which outlined methods for use in QC laboratories to control the final product and establish accept/reject limits. Over the course of the last 4 years the programme has grown to adulthood and each plant now has at least one person involved in final product analysis, 'in process' control, and monitor- ing general factory hygiene standards. These people in the majority of cases are QC chemists who took over the task because of personal interest, the minority are fundamentally more microbiologically orientated who were taken on in the course of natural turnover of staff in the QC laboratory. It is generally felt that the introduction was a challenge well met rather than a traumatic experience. As the author has said, elsewhere (1), 'Each manufacturer must assess and impose suitable standards for his product lines. Regrettably, standards will vary in an absolute sense throughout industry since the present structure of the cosmetic and toiletry industry interweaves with the manufacture of sterile and non-sterile pharmaceuticals and foodstuffs.' Throughout the plants regardless of product category the 'in house' standard imposed was, from the beginning, 100 organisms per g or per ml of product due to the fact that non-sterile pharmaceuticals and toiletties were and are manu- factured alongside each other. In the beginning all washing and disinfecting procedures were reviewed
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)