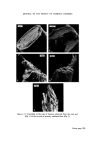
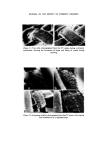
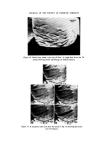
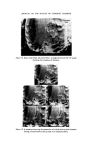
WATER-BORNE BACTERIA AND SHAMPOO SPOILAGE 279 from the column vary with the frequency with which the column is used or cleaned and disinfected (23). There is now considerable practical evidence indicating a positive correlation between high levels of bacterial contamination in water used in manufacture and the incidence of contaminated toiletry products. This is presumably because the greater the number of contaminants in the water the wider the range of resistances to unfavourable conditions and the greater the probability of bacteria being present which are capable of surviving and multiplying in the product. The range of resistance is likely to be widest where the population is made up from a range of different species of bacteria rather than large numbers of only a small number of species. It would also be theoretically possible to have very heavily contaminated water presenting no threat to product stability because of the low resistance of the bacteria or, in reverse, water containing small numbers of bacteria which, because they are well suited to growth in the product, will give rise to contamination. In order to perform Challenge Tests on shampoos in the laboratory it is often found necessary to use very large inocula, usually of the order of 106-107 bacteria per gram of product. This is, however, much higher than would normally be found as an initial inoculum during manufacture in a well-ordered production unit and product contamination is known to occur even when the level of contamination in the plant is low and water of good bacteriological quality is used. MATERIALS AND METHODS Empicol ESB 3/S (sodium lauryl ether sulphatem27.3•o active) a specially prepared preservative-free batch supplied by the Marchon Division of Albright and Wilson Ltd. Lauryl Isopropanolamide--Marchon Division of Albright and Wilson Ltd. Nutrient Agar CM3--Oxoid Ltd. Tryptone Soya Agar CM131--Oxoid Ltd. Peptone Bacteriological Neutralized L34•Oxoid Ltd. Because of the wide variety of different ingredients used all experimental shampoos are atypical. It is, however, possible to produce a model system which is representative of a general shampoo formulation. For this work the following formulation was used.
280 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Empicol ESB 3/S (sodium lauryl ether sulphate) (27.3•o active) Lauryl isopropanolamide 1.5 Sodium chloride 0.5 Sterile distilled water to 100.0 •o w/w 36.6 (equivalent to 10•o active detergent) After preparation the model shampoo was sterilized by filtering it through a 0.2 [tm filter membrane. Sodium lauryl ether sulphate (SLES) was chosen as the detergent because products containing it are known to be amongst those most suscep- tible to contamination, possibly due to its relatively low toxicity to Gram- negative bacteria. It is also widely used as a detergent in shampoos. The bacterium selected as the experimental contaminant was a strain of Enterobacter cloacae isolated from a contaminated shampoo. We have found this organism to be particularly resistant to anionic detergents. Three 16 oz nutrient agar slopes were each inoculated with 1 ml of a 24 h nutrient broth culture of Enterobacter cloacae and incubated at 28 ø overnight. The bacteria were washed from the slopes using distilled water and washed three times with water after centrifugation. The bacteria were finally re-suspended in distilled water and stored at 4 ø for 4 days before use. Total viable counts Total viable counts on bacterial suspensions and inoculated shampoos were performed in Tryptone Soya Agar using the pour-plate method with 0.1 •o peptone water as the diluent. Total viable counts on mains water were performed in Nutrient Agar using sterile mains-water as the diluent. In each case the plates were incubated at 28 ø for 7 days before counting. Influence of inoculum size on the survival and multiplication of bacteria in a shampoo Total viable counts were performed on the stored suspension of E. cloacae. A series of decimal dilutions of the suspension were then made in distilled water so as to give the following range of concentrations of bacteria: 109, 108, 107, 10 ø, 105, 104, and 10 a ml -•. 0.2 ml of each suspension was added to duplicate 20 g amounts of sterile shampoo. The inoculated shampoo was incubated at 28 ø and total viable counts were performed at intervals up to 14 days.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)