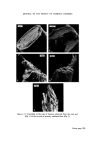
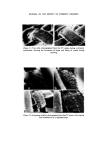
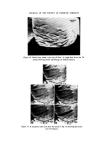
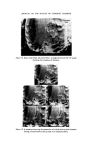
308 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS populations to provide meaningful information. It can be seen that 17•o of the hair of the female population can be described as blonde, whilst for the male population only 1 •o of male hairs were blonde. In group 6, six out of seven women had blonde hair as a result of chemical bleaching whereas, not surprisingly, none of the male population had bleached hair. One of the six females in group 7 had bleached hair, whilst one in four females in group 8 had bleached hair. Hence it will be obvious that a blonde, bleached hair is more likely to be female than male. A blonde female unbleached hair repre- sents only 1 •o of the female population, and therefore it may be concluded that hair lighter than group 5, i.e. 6, 7, 8 can provide useful information. Identification of hair cosmetic treatments Bleaching The use of commercially available bleaches to lighten hair brings about the bleaching of the hair pigment (melanin), together with structural damage to the tt-keratin (hair protein). The cuticular layer containing the tt-keratin is gradually abraded away due to the movement of individual hairs across one another and by brushing and combing. The extent of the damage to single fibres can be estimated by observation of the uptake of dye from an aqueous solution of methylene blue. The amino-acid composition of bleached hair is not greatly different from unbleached hair, with the exception that, in the former, a high proportion of the cysteine and cystinc is converted into the corresponding sulphonic and cysteic acid. It is the pre- sence of the anionic sulphonic acid groups which is responsible for the up- take of methylene blue (a cationic dye). It is apparent from the present work that the degree of hair damage is closely related to the extent of hair bleach- ing. For a given bleached fibre the uptake of methylene blue is greater at the tip of the fibre and least near the root. It has been established that if 1 cm lengths of hair are obtained from the same head but from different hairs and from the same distance from the root, then methylene blue uptake is similar in each case. The methylene blue test is therefore a useful compara- tive test for assessing hair damage caused by chemical bleaching. No other cosmetic treatments have been found to interfere with the methylene blue reaction. The occurrence of bleached hair for the different colour groups is shown in Table II. Technique. A 1 cm portion of hair is fixed to a cavity slide using perspex cement or cellulose acetate, and several drops of aqueous methylene blue solution (0.5•o) added to completely cover the hair. After 4 min the hair is
HUMAN HEAD HAIR AS FORENSIC EVIDENCE 309 Table II. Occurrence of bleached hairs Colour group (female) 1 2 3 4 5 6 7 8 9 % population (100) 0 28 20 9 14 7 6 4 12 No. hairs bleached 0 0 0 0 0 6 1 1 0 rinsed with water and observed. Extensive hair damage is indicated by intense blue/violet colouration. Unbleached hairs do not absorb any dye under these conditions. Hair lacquer residues A technique has been developed (Crockett (29)) for the examination of lacquer residues by infrared spectroscopy. This technique enables the type of resin used to be identified and requires only 2-3 cm of hair. However, before this technique can be used some data on the use of lacquer in the population is required. Polyvinyl pyrollidone (the major constituent of many hair lacquers) is, however, the basic material for a wide range of hair cosmetic preparations other than lacquers (e.g. sets, conditioners, etc). Hair dyes Hair dyes can be broadly classified into three types: Temporary rinses. These are essentially coloured pigments which are dis- solved in water and absorbed on the surface of the hair. They contain a wide range of compounds including azo-dyes, triphenylmethane dyes and anthraquinones. Semi-permanent dyes. These are frequently nitroaromatic amines which are strongly absorbed on the surface of the hair and give a good depth of shade. Permanent or oxidation dyes. Permanent dyes are mostly based on p-phen- aminediamine. In the dyeing process the amine penetrates the hair shaft and enters the cortex, where it is oxidized with hydrogen peroxide to yield a complex polymeric product. The development of a thin-layer chromatographic system to identify these types of dyes on single fibres has been studied (Logan (30)) but found to be impractical due to great similarity between the polymerized product of different manufacturers. Where sufficient fibres are available (i.e. 50 cm) the dyes may be extracted and identified by TLC. However, a considerable
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)