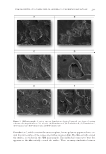
JOURNAL OF COSMETIC SCIENCE 170 At the end of the loading procedures, the encapsulation effi ciency of the SLNs was 49.22%. Hydrophilic substances such as caffeine tend not to dissolve in the lipid matrix of SLNs. Therefore, such active agents are expected to be poorly encapsulated in SLNs (23). In vitro release studies were performed and Figure 4 shows the cumulative release profi le of Caf from Caf-SLNs. The release profi le demonstrated a two-step release behav- ior an initial burst released without detectable lag time followed by a relatively slow release. The initial burst release in the fi rst 5 min was nearly 20% of the total amount of Caf followed by a sustained release of the remaining Caf over nearly 6 h (Figure 4). The initial burst of Caf release from the Caf-SLNs may originate from the Caf molecules clos- est to the SLN surface. The sustained release phase may be related to the diffusion of Caf molecules from the lipid matrix. In addition, it may be because of the effect of decreasing Caf amounts. Such release behavior also has been observed in the release of Caf from nio- somes by Khazaeli et al. (15). The release kinetics of the Caf-SLNs were evaluated using zero-order, fi rst-order, Higuchi, Hixson–Crowell, and Korsmeyer–Peppas equations. The results of the calculations indicated that the release profi le of the Caf from Caf-SLNs best fi t with the Korsmeyer–Peppas model release kinetics (Table 3). The n value was between 0.5 and 1.0, which indicated that the release mechanism was anomalous transport. The release kinetic model indicated both diffusion and erosion release patterns from polymeric Figure 2. (A) SEM micrographs of caffeine, (B) caffeine loaded SLNs, and (C) unloaded SLNs.
STUDIES OF CAFFEINE-LOADED SOLID LIPID NANOPARTICLES 171 type carriers (24). Thus, it was named anomalous drug transport. The kinetic results show that Caf release was diffusion controlled and, because of lipid matrix degradation, lipid matrix erosion controlled. CONCLUSIONS A new delivery system using SLNs with Caf were successfully formulated and character- ized for the fi rst time. DLS and SEM measurements clearly indicated that Caf-SLNs had a spherical structure and nanometric particle size with good PI values. Caf was loaded into the SLNs with reasonable encapsulation effi ciency. The release profi le of Caf from the Figure 4. The cumulative release pro fi le of caffeine from Caf-SLNs. Figure 3. Thermograms of unloaded SLNs, caffeine, caffeine-loaded SLNs, and Softisan 100® obtained using DSC.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)