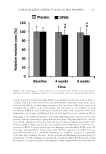
JOURNAL OF COSMETIC SCIENCE 282 values were 11.53 ± 2.41 and 10.04 ± 2.73 g/m2/h, respectively (p 0.05 vs. both pla- cebo and baseline at the two time points). Figure 1C and D illustrate changes of ski n elasticity and fi rmness at three time points of the study. At baseline, the mean values of skin elasticity were 0.84 ± 0.08 and 0.84 ± 0.09 AU of placebo and DPKE creams, respectively after 4 weeks, these values were 0.85 ± 0.07 and 0.89 ± 0.06 AU, respectively (p 0.05 vs. placebo and p 0.01 vs. base- line), and after 8 weeks, the values were 0.86 ± 0.09 and 0.94 ± 0.10 AU, respectively (p 0.001 vs. both placebo and baseline). At baseline, the mean fi rmness in the DPKE test skin was 0.27 ± 0.08 AU, and after 4 and 8 weeks, the fi rmness had diminished to 0.23 ± 0.06 and 0.21 ± 0.06 AU, respectively (p 0.01 vs. placebo). Figure 1E illustrates changes of skin melanin density at three time points of the study. At baseline, the mean readings of skin melanin were 273.8 ± 27.9 and 265.6 ± 26.4 AU of placebo and DPKE creams, respectively. After 4 weeks, these values were 267.4 ± 30.2 and 260.8 ± 27.5 AU, respectively, and after 8 weeks, the values were 275.2 ± 28.4 and 255.3 ± 26.5 AU, re- spectively (p 0.05 vs. both placebo and baseline). There were no signifi cant changes in erythema levels for both placebo and DPKE creams over 8 weeks (Figure 1F). ANTERA 3D® MULTISPECTRAL ANALYSIS The signifi cant reduct ion in the wrinkle depth compared w ith pretreatment values was confi rmed by objective analysis with the Antera 3D® analyzer performed at baseline, and after 4 and 8 weeks of treatment. Several wrinkle measurements showed signifi cant im- provements after 4 and 8 weeks compared with both baseline values and placebo cream. With the medium fi lter, there was a signifi cant decline in the overall size after 4 and 8 weeks by 18.4% and 28.6%, respectively (from 56.4 ± 22.2 to 46.0 ± 21.5 (p 0.05 vs. baseline and placebo), and 40.3 ± 22.1 AU (p 0.01 vs. baseline and placebo, paired t-test, Figure 2A). The maximal depth was signifi cantly decreased by 22.5% and 29.6%, respectively (from 0.410 ± 0.19 mm to 0.322 ± 0.18 mm and 0.284 ± 0.23 mm, respec- tively (p 0.05 vs. baseline and placebo for all, paired t-test, Figure 2B). Moreover, sig- nifi cant improvements in the indentation index and skin roughness index were also evident after 4 and 8 weeks however, the reductions of these parameters were more pro- nounced after 8 weeks, compared with baseline and placebo levels (p 0.01 and p 0.001, respectively, Figure 2C and D). The overall reduction in the skin color was also Table III Clinical Assessment Rated on a Three-Point Scale Baseline After 8 weeks Placebo DPKE Skin roughness score 2.24 ± 0.81 2.17 ± 0.64 1.76 ± 0.94**,# Skin texture homogeneity score 2.18 ± 0.76 2.01 ± 0.90 1.55 ± 0.66***,# Skin melanin pigmentation score 2.36 ± 0.95 2.28 ± 0.74 1.90 ± 0.84*,# Skin redness score 2.04 ± 0.84 2.11 ± 0.72 1.93 ± 0.93 Data are presented as mean ± SD of 36 subjects. Signifi cance levels: *p 0.05, **p 0.01, and ***p 0.001 versus baseline. #p 0.05 versus placebo (ANOVA).
DATE PALM KERNEL EXTRACT ON FACIAL SKIN WRINKLES 283 evaluated during the follow-up visits using the Antera 3D. Multispectral analysis of the skin color showed that DPKE cream statistically reduced the average concentrations of skin melanin at the two time points of assessment compared with both the baseline and placebo measurements (p 0.05 for all, Figure 3). The placebo cream had no signifi cant effects on the overall wrinkle size, depth, indentation, and roughness indices or skin Figure 1. Biop hysical m easurements in 36 subjects at baseline and after 4 and 8 weeks of topical treatment with 5% DPKE cream. (A) Skin hydration, (B) TEWL, (C) skin elasticity, (D) fi rmness, (E) melanin pigmen- tation, and (F) erythema. Data are presented in AU as mean ± SD. Signifi cance levels: *p 0.05, **p 0.01, and ***p 0.001 versus baseline. #p 0.05, ##p 0.01, and ###p 0.001 versus placebo (ANOVA).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)