83 ENCAPSULATED TTO IN FACIAL CREAMS (30) T. Adegoke and D. Arotupin, Preservative capacity of some commercial cosmetics in Akure Metropolis, Nigeria, J. Adv. Microbiol., 5(2), 1–11. (31) M. B. Oliveira, D. C. M. Vieira, T. M. Fiúz, H. R. N. Salgado, and M. Chorilli, Microbiological control of moisturizing mask formulation added of hibiscus flowers, assai palm, black mulberry and papaw glycolic extracts, Int. J. Pharm. Pharm. Sci., 5(1), 342–345 (2013). (32) H. Lambers, S. Piessens, A. Bloem, H. Pronk, and P. Finkel, Natural skin surface pH is on average below 5, which is beneficial for its resident flora, Int. J. Cosmet. Sci., 28(5), 359–370 (2006). (33) T. Mitsui, “Stability of cosmetics,” in New Cosmet. Sci., T. Mitsui. Ed. (Elsevier Amsterdam, 1997), pp. 191–198. (34) R. Campana, C. Scesa, V. Patrone, E. Vittoria, and W. Baffone, Microbiological study of cosmetic products during their use by consumers: health risk and efficacy of preservative systems, Let. Appl. Microbiol., 43(3), 301–306 (2006). (35) ISO International Organization for Standardization. ISO 22717:2015, Cosmetics — Microbiology — Detection of Pseudomonas aeruginosa. In: (2015). (36) ISO International Organization for Standardization. ISO 22718:2015, Cosmetics — Microbiology — Detection of Staphylococcus aureus. In: (2015). (37) ISO International Organization for Standardization. ISO 16212:2017, Cosmetics — Microbiology — Enumeration of yeast and mould. In: (2017). (38) J. Behravan, F. Bazzaz, and P. Malaekeh, Survey of bacteriological contamination of cosmetic creams in Iran (2000), Int. J. Dermatol., 44(6), 482–485 (2005). (39) D. K. Brannan and P. A. Geis, Cosmetics microbiology. In: Schaechter M, ed. Encyclopedia of Microbiology (3rd Edition), Cambridge: Academic Press. 270–280 (2009). (40) H. Zeitoun, M. Kassem, D. Raafat, H. AbouShlieb, and N. Fanaki, Microbiological testing of pharmaceuticals and cosmetics in Egypt, BMC Microbiol., 15, 275–275 (2015). (41) I. T. Some, P. Bogaerts, R. Hanus, M. Hanocq, and J. Dubois, Improved kinetic parameter estimation in pH-profile data treatment, Int. J. Pharm., 198(1), 39–49 (2000).
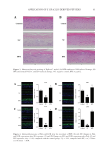
84 Address all correspondence to Bingliang Wang, wangbl@usx.edu.cn Junxiang Li, lijunxiang@acrdc.cn Application of Euglena gracilis-Derived Peptides as a Cosmetic Ingredient to Prevent Allergic Skin Inflammation XUHUI LI, CHUNXIN XIA, DANNI KONG, MING XU, JIN ZHU, CONGFEN HE, BINGLIANG WANG AND JUNXIANG LI School of Life Science, Shaoxing University, Shaoxing, Zhejiang, 312000, China (D.K., B.W.) Zhejiang Provincial Key Laboratory of Applied Enzymology, Yangtze Delta Region Institute of Tsinghua University, Zhejiang, China (X.L.) AgeCode R&D Center, Yangtze Delta Region Institute of Tsinghua University, Zhejiang, China (X.L., C.X., M.X., J.Z., C.H., J.L.) TUS-HARVEST biotech. Co. Ltd., China (C.X., M.X., J.Z., C.H., J.L.) Beijing Key Laboratory of Plant Resources Research and Development, School of Science, Beijing Technology and Business University, Beijing, China (C.H.) Accepted for publication January 18, 2022. Synopsis Bioactive peptides have a wide range of therapeutic activities. They have emerged as interesting candidates for cosmetic ingredients. Euglena gracilis contains high amounts of proteins consisting of all 20 proteinogenic amino acids. As a result, it is thus proposed that enzymatic peptides from E gracilis proteins would have multiple biological activities. To verify their potential application in cosmetics, the therapeutic effects of E gracilis-derived peptides (BPE) on allergic inflammation were evaluated in this study. In 2D skin models, HaCaT cells were used to assess cell viability, a fluorescence of 2’,7’-dichlorofluorescein diacetate was used to evaluate reactive oxygen species (ROS) contents, and an enzyme linked immunosorbent assay (ELISA) analysis of tumor necrosis factor α (TNF-α) reflected inflammatory response. Meanwhile, transcriptome analysis was performed to reveal the changes in gene expression. In 3D skin models, hematoxylin-eosin staining revealed the barrier-repairing ability of BPE. Filaggrin (FLG) and loricrin (LOR) levels were measured by their immunofluorescence. ELISA analysis of thymic stromal lymphopoietin (TSLP) also reflected the inflammatory response. A double-blind clinical trial was conducted to evaluate the erythema and itch, which reflects the degree of inflammation. Sodium lauryl sulfate, lipopolysaccharide, and histamine were used to induce allergic responses in vitro and in vivo. In 2D skin models, BPE increased cell viability from 41 to 55%. It also decreased intracellular ROS content from 100% to 61%. Combined with the decrease in TNF-α expression, it showed that BPE exerted multiple biological activities, including antioxidant and anti- inflammatory activities. According to the transcriptome analysis, BPE significantly changed the amounts of inflammation– and immune–response-related genes and enhanced the anti-inflammatory ability of cells. In 3D skin models, hematoxylin-eosin staining showed that BPE repaired the damages induced by Poly I:C and lipopolysaccharides. After BPE treatment, filaggrin and LOR expressions reached 124 and 127% J. Cosmet. Sci., 73, 84–95 (March/April 2022)
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)