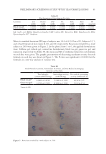
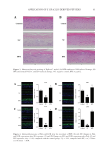
85 APPLICATION OF E GRACILIS-DERIVED PEPTIDES of the negative control, respectively, and the TSLP content significantly increased. It suggested that BPE treatment significantly alleviated inflammatory response and repaired the inflammation-induced damages. Furthermore, clinical trials in vivo also showed that BPE significantly suppressed allergic inflammation. The simulative experiments in 2D and 3D skin models as well as clinical trials verified that BPE had therapeutic effects on allergic inflammation. Our results propose BPE as a novel cosmetic ingredient to prevent allergic responses. INTRODUCTION Bioactive peptides are specific protein fragments, usually ranging from 2 to 20 units, with a molecular weight under 3 kDa. They can interact with a large number of biological macromolecules and biochemical compounds, showing a wide range of therapeutic antimicrobial, antihypertensive, antioxidant, antitumoral, antiproliferative, hypocholesterolemic, and anti-inflammatory activities (1,2). Bioactive peptides are generally obtained from diverse natural protein sources, such as food protein, vertebrates, bacteria, and microalgae (3). Among these protein sources, the microalgae-derived peptides are advantageously employed as bioactive compounds in cosmetics as they are selective, effective, safe, and well-tolerated once consumed (1). Euglena gracilis is a group of free-living, single-celled flagellates (4). It belongs to the green plant line, but in evolutionary terms it is most closely related to the protozoa (5). Nowadays, it is seen as one of the most attractive algal species for bioproducts. The most important aspect of E gracilis is that it contains high amounts of proteins, vitamins, and polyunsaturated fatty acids (6,7). Euglena gracilis has a relatively high amino acid content and higher nutritional quality compared to Chlorella and Spirulina (8). E gracilis contains all 20 proteinogenic amino acids (9). Although E gracilis has been exploited for decades (7), no studies focus on the application of bioactive BPE. According to previous studies of other microalgae-derived bioactive peptides, E gracilis are effective in promoting skin hydration and in fighting skin osmotic stress damages. Moreover, they can abolish reactive oxygen species (ROS) induction caused by oxidative stress agents (10). Because of this, it is interesting to investigate the skin care-related activity of bioactive peptides derived from E gracilis. The skin is one of the largest immunologic organs and is often a target for allergic and immunologic responses (11). Both external and internal factors can induce immune- mediated responses including dermatitis, urticarial, and angioedema, among others. Due to occupational exposures, face and hands suffer from a high incidence of dermatitis (12). Except for the clinical practice, cosmetic interventions are required to avoid the deterioration of quality of life (13). Some topical agents, such as zinc salts, taurine, and D-panthenol, can create a protective barrier over the skin and reduce the impact of external irritants, resulting in an effective treatment for rashes (14). However, it is still unclear whether bioactive peptides, especially the E gracilis-derived peptides (BPE), can be a promising alternative as therapeutic agents against allergic skin inflammation. To verify this previously mentioned hypothesis, we performed a variety of biological experiments based on 2D skin models, transcriptome analysis, and 3D skin models, which demonstrated that BPE reduced allergic inflammation.
86 JOURNAL OF COSMETIC SCIENCE MATERIALS AND METHODS PREPARATION OF BIOACTIVE PEPTIDES Euglena gracilis (FACHB-848) was purchased from the Freshwater Algae Culture Collection at the Institute of Hydrobiology (Wuhan, China) and then cultured in an autotrophic medium (AF-6) with a controlled temperature and light exposure (25°C, light/dark =12/12, 30 μm photons m−2 s−1) (15). In the exponential phase, E gracilis cells were collected and washed with distilled water. Cells were then broken by an ultrasonic method, and its pH was adjusted to approximately 7.0–8.0. The crude extract was centrifuged at 4°C with 10,000 g for 30 min. The protein was precipitated at pI. The pH of the supernatant was then briefly decreased to 4.0 progressively using hydrochloric acid (16). The protein precipitation was collected by centrifugation and redissolved in double distilled water at pH of 7.0. After determining the protein concentration using the Coomassie brilliant blue method (0.54 mg/mL), the protein solution was hydrolyzed by a commercial neutral protease (Beijing Solarbio Science &Technology Co., Ltd., Beijing, China). The hydrolysis process was conducted with a 0.1% neutral protease under 50°C for 4 h. The enzymatic peptides were then ultrafiltered with a 3 kD ultrafiltration membrane and stored at −80°C for the next experiments. PREPARATION OF CELLS AND 3D EPIDERMAL SKIN MODEL HaCaT and RAW 264.7 cells, purchased from Fenghui Biology (Hunan, China), were maintained in Dulbecco’s modified Eagle medium (DMEM) solutions (Wisent, China), which were supplemented with 10% (vol/vol) fetal bovine serum (Invitrogen, USA) and 1% penicillin/streptomycin (Lanke Biology, China, 100 IU/mL). Primary culture cells (HDF), purchased from Beina Biology in China, was also cultured in low-glucose DMEM solutions (Wisent, China), which were also supplemented with 10% fetal bovine serum (Gibco, Grand Island, NY) and 1% penicillin/streptomycin (Lanke Biology, Hangzhou, China, 100 IU/mL). 3D epidermal skin model Epikutis® (Zhengzhou Biocell Biotechnology Co., Ltd. Henan, China) was maintained in EpiGrowth solutions. Cell and Epikutis® were cultured at 37°C in a 5% CO 2 -containing humidified atmosphere. ASSAY OF CELL VIABILITY Immortalized keratinocytes (HaCaT cells) were cultured in 96-well plates for 24 h at 4 × 104 cells/mL (100 μL/well). Then, HaCaT cells were treated with 100 mg/L of sodium lauryl sulfate (SLS, Sigma, USA) and supplemented with 1% BPE for 24 h. The positive controls were treated with 0.1% of Comthing® SGS (ComThings SAS, Immeuble Premium, Nice, France), a commercial product (http://greaf-cc.ecer.com/sale-9601118-comthing-sgs.html). After incubation, HaCaT cells were washed by PBS three times, and cell viability was determined using the Cell Counting Kit-8 Assay reagent (Dojindo, Japan). ASSAY OF REACTIVE OXYGEN SPECIES (ROS) Human dermal fibroblasts (HDF cells) were cultured in 96-well plates at 1 × 104 cells/mL (100 μL/well) for 48 h. The cells were then treated with 10 μm of hydrogen peroxide (H 2 O 2 ,
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)