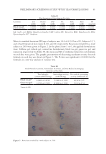
87 APPLICATION OF E GRACILIS-DERIVED PEPTIDES Sigma, USA) for 2 h. H 2 O 2 -treated cells were washed twice with PBS and incubated in DMEM solutions for 24 h. The positive controls were treated with 100 μg/mL of Vitamin E and experimental groups were treated with 5 μg/mL of BPE. For ROS analysis (17), 10 μm of 2’,7’-dichlorofluorescein diacetate (DCFH-DA, Beyotime, China) was added in the cell suspension at 37°C for 30 min in darkness. The intensity of fluorescence was measured with a fluorescent microplate reader (Molecular Devices, USA). ASSAY OF INFLAMMATORY RESPONSE The Mouse tumor necrosis factor α (TNF-α) enzyme linked immunosorbent assay (ELISA) Kit (Multi, China) and Human thymic stromal lymphopoietin (TSLP) ELISA Kit (Boster, China) were used to measure the content of extracellular TNF-α and TSLP (18,19). In brief, RAW 264.7 cells were cultured in 96-well plates at a density of 2 × 104 cells per well. After 24 h of cultivation, 1 mg/L of lipopolysaccharides (LPS) (Sigma, USA) was used to stimulate inflammatory response of cells. Meanwhile, the experimental groups were supplemented with 1% BPE for 24 h, and the positive controls were treated with 100 μg/mL of dexamethasone (DXMS). The extracellular medium was then collected to determine TNF-α. The absorbance value was detected at 450 nm, and the enzyme activity of TNF-α in the sample was calculated according to a standard curve. The culture medium of Epikutis® was also collected to analyze the inflammatory response. TSLP was measured using Human TSLP ELISA Kit at 450 nm with a microplate reader (BioTek, USA). To explore the changes in gene expression, transcriptome analysis was conducted. LPS- induced RAW 264.7 was briefly incubated with BPE in 6-well plates for 24 h. After being washed by 1× phosphate-buffered saline, RNA was extracted with a TRIzol reagent (Invitrogen, USA) according to the manufacturer’s instructions. cDNA was then synthesized and amplified to construct sequencing libraries using LianChuan Biology (Hangzhou, China). Graphene oxide enrichment analysis referred to the website David (https://david. ncifcrf.gov/summary.jsp), and the heatmap was analyzed via the website Morpheus (https:// software.broadinstitute.org/morpheus/). 3D EPIDERMAL SKIN MODEL The 3D epidermal skin model (Epikutis®) was cultured in 6-well plates with EpiGrowth solutions. After treatments with Poly I:C (Sigma, USA), LPS (Sigma, USA), and SLS (Sigma, USA), Epikutis® was fixed with 4% polyformaldehyde for 24 h. Afterward, the samples were dehydrated, embedded in paraffin, and cut into 5 μm sections. After hematoxylin and eosin staining, the sections were dried at room temperature and then mounted with neutral gum. Finally, the morphology of Epikutis® was examined under a microscope (Leica, German) (19). Immunofluorescence analysis was performed as follows (20): the paraffin sections were put into xylene for 10 min and hydrated with anhydrous and 95% ethanol after rinsing with distilled water, the sections were put into 0.01 M of sodium citrate with high pressure and supplemented with one drop of 3% H 2 O 2 for 30 min, which could block the activity of endogenous peroxidase the sections were then incubated with anti- filaggrin (FLG) or anti-loricrin (LOR) (Abcam, USA) at 4°C overnight anti–Mouse-488 (Abcam, USA) was
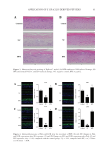
88 JOURNAL OF COSMETIC SCIENCE used as a secondary antibody for 1 h of incubation and the sections were finally observed by fluorescence microscopy (Leica, German). Analysis of immunofluorescence intensity was performed in a blinded manner. CLINICAL TRIALS A double-blind clinical trial was conducted to evaluate the changes in allergic inflammation. The clinical trial consisted of 16 volunteers aged 20–58 years old (4 males and 12 females). In brief, 4 cm2 areas on the surface of one forearm was provoked with 10 mg/mL of histamine dihydrochloride. After provocation, double distilled water and a 5% BPE solution were gently rubbed on the targeted area. Erythema, which reflects the degree of inflammation, was evaluated by a Cutometer Dual MPA 580 with a probe Colorimeter CL 400 (Courage and Khazaka Electronics, Cologne, Germany) and expressed in the L*a*b* system. The a* value on the green–red axis was used to assess the erythema. All measurements were performed under controlled environmental conditions (room temperature of approximately 20.1–22.8°C and an average relative humidity of approximately 41.7–60.4%). All patients underwent subjective evaluation of their itch using a customer questionnaire (21). STATISTICAL ANALYSIS All data were presented as the mean of three individual experiments. Significant differences were calculated using student’s t-test via GraphPad Prism 8 software. Differences were considered statistically significant when p values 0.05. RESULTS AND DISCUSSION 2D SKIN MODELS Allergic inflammation was simulated through chemical induction in 2D skin models. The efficacy of BPE for dermatitis treatment was evaluated based on these models. HaCaT cells were chosen to examine the cell protection function of BPE (22). Figure 1A illustrated that cell viability significantly decreased after SLS exposure. The negative control reached 41%, but it was improved by the treatments with anti-inflammation pharmaceuticals and BPE, which were 62 and 55% respectively. ROS level is an important indicator during oxidation reaction (23,24). Figure 1B showed that ROS levels were 41 and 68% for the vitamin E and BPE treatments. The antioxidant activity of BPE is similar to other algal peptides (25). It implied that BPE performed a potential function in alleviating oxidative damages. Because of this, the results indicated that BPE played an effective role in the process of cell protection. Expression of TNF-α is sensitive to inflammatory response (26,27). Changes in TNF-α contents were analyzed to evaluate inflammatory response. As shown in Figure 1C, LPS exposure significantly increased the TNF-α content of RAW 264.7 cells, which indicated a remarkable inflammatory response. After DXMS and BPE treatments, TNF-α contents significantly decreased (Figure 1C), indicating BPE diminished inflammation. In all, 2D skin models verified that BPE exerted antioxidant and anti-inflammatory activity.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)