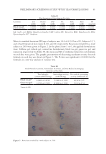
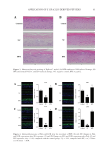
91 APPLICATION OF E GRACILIS-DERIVED PEPTIDES Figure 3. Hematoxylin-eosin staining of Epikutis® model. (A) BPE-ameliorated SLS-induced damage (B) BPE ameliorated Poly I:C and LPS-induced damage. NC: negative control BPE: 5 μg/mL. Figure 4. Immunofluorescence of FLG and LOR after the treatment of BPE. (A) and (B) Changes in FLG and LOR expressions after SLS exposure. (C) and (D) Changes in FLG and LOR expressions after Poly I:C and LPS exposures. ##p 0.01, compared with the control group **p 0.01, compared with NC, n =3. Values are the mean ± SEM.
92 JOURNAL OF COSMETIC SCIENCE CLINICAL EVALUATION A double-blind trial was performed to evaluate BPE’s clinical effects on the allergic inflammation. Forearms of 16 adults were provoked with 10 mg/mL of histamine dihydrochloride, which simulated symptoms of allergic dermatitis. As shown in Table I, the decrease in average a* values reached 8.66 after BPE treatment, while the control Figure 5. Analysis of TSLP level by ELISA. DXMS: dexamethasone ##p 0.01, compared with the control group *p 0.05, compared with NC, n =3. Values are the mean ± SEM. Table I Changes in Erythema (a* Value) After BPE Treatment Compared With Control Groups (Double Distilled Water)a,b Double distilled water BPE Treatment Time 0 min 10 min 30 min 60 min 0 min 10 min 30 min 60 min Maximum 39.22 42.37 39.6 40.87 42.51 47.27 42.3 42.34 Minimum 17 16.16 17.12 15.53 17.42 17.92 17.75 17.65 Average 30.95 29.66 26.5 23.84 32.42 30.3 26.2* 23.76* Standard Deviation 7.79 7.82 7.12 6.69 7.68 7.16 6.5 6.01 Δa* Value – −1.29 −4.45 −7.11 – −2.11 −6.22 −8.66 a n =16. b *p 0.05. Table II Changes in Itch Intensity After BPE Treatmenta,b, Compared With Control Groups (Double Distilled Water) Double distilled water BPE Treatment time 0 min 10 min 30 min 60 min 0 min 10 min 30 min 60 min Maximum 4 4 3.5 3 4 4 3.8 3 Minimum 3 2 1 0 2.5 2 0.5 0 Average 3.7 3.23 2.36 0.97 3.51 2.86 1.73 0.53 Standard Deviation 0.43 0.84 0.9 0.89 0.56 0.78 0.9 0.88 Δ Value – −0.47 −1.34 −2.73 – −0.65 −1.78 −2.99 a n =16, data were collected from population survey based on the double-blind trial. b *p 0.05.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)