354 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS applied after ascertaining that there was no inflammation on the part to be tested. Blood samples were collected from the ear lobe by in- travenous puncture and assayed for vitamin B0 content. The composition of the hydrophilic ointment is as follows: Isopropyl myristate 1 g. Cetanol 0.25 g. Stearic acid 0.4 g. Paraffin wax 1 g. NIKKOL BL-9* 0.2 g. NIKKOL BC-5t 0.15 g. Mineral oil 1.35 g. 10% Triethanolamine solution 1 ml. Pyridoxine derivative 1 • Water to make 1 • g. In the preparation of the ointment containing pyridoxine hydro- chloride, the triethanolamine solution was omitted from the above formulation. The vitamin B6 content in blood was determined as follows: Standard curve: In the case of pyridoxine hydrochloride and of pyridoxine-3,4-dibutyrate, 1 ml. of an aqueous sample solution of known concentration (about 0.5 m/mi.) was acidified with 6 mi. of 15% H2SO4, and the solution was heated at 100øC for one hour. After cooling, the pH of the solution was adjusted to 5.4, and the volume made up to 50 ml. with water. Samples of this solution (0.25, 0.5, 1 and 2 mi.) were pipetted into test tubes and diluted with water to 2.5 mi. After adding 2.5 mi. of culture medium and steriliz- ing at 100øC for 15 minutes, 1 drop of preincubated suspension of Saccaromyces Carlsbergensis culture was added, and the solution was incubated at 30 øC for 20 hours. Absorbancy was then measured at 610 mu, and a standard curve was constructed. For the construction of the standard curve for pyridoxine-3,4- dioctanoate, 1 mi. samples of ethanolic solutions were hydrolyzed as mentioned above. Pyridoxine assay in blood: A mixture of 1 g. of blood and 6 mi. of 15% H2SO4 was heated at 100 øC for one hour. After cooling, the pH of the solution was adjusted to 5.4, and the liquid was centrifuged to remove blood pigments. The supernatant solution was separated, and its volume was adjusted to 50 mi. with water. The microbio- * Polyoxyethylene lauryl alcohol ether. Nikko Chemicals Co., 1, 1 chome, Nihonbashi Bakurocho, Chiyoda-ku, Tokyo. t Polyoxyethylene cetyl alcohol ether. Nikko Chemicals Co.
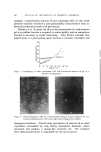
PYRIDOXINE-3,4-DIACYLATES IN COSMETICS 355 logical assay was carried out similarly as mentioned above, and the amount of pyridoxine was determined by reference to the standard curves. The results are shown in Fig. 2. Pyridoxine and pyridoxine-hydro- chloride were absorbed immediately, but their content in blood de- creased rapidly. In contrast, the dibutyrate was also absorbed im- mediately, but the blood concentration remained at a constant level. The absorption of the dioctanoate and di-iso-octanoate was delayed, but the blood concentration increased gradually. The absorption of dilaurate and dipalmitate was not studied in this experiment. These 1000' -• 8oo. •o rldorlne • 600- o• Pyrldo lbutMrat " • 400- • /• • Pyridoxlne Dl-I sonic tano • te • 200- • PyrldoxlnDiootan ?lme ( hrs. ) Figure 2. Percutaneous absorption of pyridoxine and its derivative (1% in 0/W cream base) facts might suggest that cosmetics or ointments containing intermediate chain fatty acid diester of pyridoxine maintain higher levels of vitamin B• in the living body for a longer period of time than products containing pyridoxi•e. Hydrolysis of Diacylates in Organs (•) Liver, kidney, intestine, and blood of the mouse were used in this experiment. Liver, kidney, and intestine, from which as much blood as possible was removed, were washed with water. One gram of liver, 0.4 g. of kidney, 0.7 g. of intestine and 0.4 ml. of blood were homogenized with Tyrode's solution. These homogenates were poured into 50 ml. Edenmeyer flasks, and the contents were adjusted to 7 g. with Tyrode's
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)