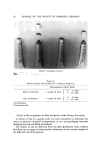
76 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS microorganisms, insufficient cleansing of the filling machine, contamination of the containers, and by the use of unskilled and careless workers. Preventing Microbial Contamination of Raw Materials A. Sterilization of Deionized Water Based upon the contamination noted in the raw materials, we considered various methods of sterilization for those raw materials, which had previously caused particular trouble. Table II shows these results, which were obtained using the sterilizer shown in Fig. 2. As deionized water is considered to be one of the most important raw materials used in the formulation and manufacture of cosmetic products, we adopted the uv sterilization treatment for the deionized water, which is used widely in other in-plant general purpose applications. B. Sterilization of Cellulose Gums The cellulose gums, used as raw materials, are often found to be con- taminated and are sources of contamination in the finished product. They may be readily sterilized by ethylene oxide gas, and under these conditions it is possible to get good sterilization. Moreover, sterilization by the ethylene oxide gas method results in a product having no noticeable change in quality and does not affect the end product. These results are shown in Table III. Prevention o[ Microbial Contamination During the Manu[acturing Process A. Microbially Free Condition During Manufacturing In consideration of a microbially free condition, processing equipment such as kettles, pipes, and cooler, etc., were automatically cleansed in a closed system, and a standardized operational method of cleansing was set up. All processing equipment (before each pieces' use in the manufacturing process) xvas sterilized as is indicated in Table IV. In order to prevent microorganisms from contaminating the kettle, all the air which was drawn into the kettle was filtered by a highly efficient Flow (Kg/hour) Table II Sterilization" of Deionized Water by uv _ Untreated Water Treated Water Microorganism count F]oxv Microorganism count (cells / ml) (Kg/hour) (cells/ml) 2400 0 2100 0 6400 0 6000 0 8800 0 10000 0 10000 0 Before uv 3.6 X 10 • Before uv 6 X 10 •Sterilizer employed: Steroline Sterilizer: Steroline System Co., Model C-4-S, Capacity: 10T/hour.
MICROBIAL QUALITY CONTROL 77 Figure 2. Uv sterilizer Table III Sterilization of Cellulose Gums by Ethylene Oxide Gas' Lot Number Microorganism Count (cells/g) Untreated EO Gas Treatment 23666 1.6 X 10 .0 0 26264 1.2 X 10 • 0 'EO gas treatment conditions: Gas density-----20 per cent (diluted with carbon dioxide) Pressure, 1.0 Kg/cn½ Temperature, 50-55øC Time, 4 hours. microporous filter mounted on the air inlet. This filter has the capability to filter out any foreign matter larger than 0.08 /•m, thereby preventing or- ganisms from entering the u,fit. The storage tank is also equipped with a microporous filter and as is shown in Fig. 3. B. Microbially Free Condition During Filling Operation 1. Clean Room: It is well known that a clean room must be utilized in order to obtain a microbially free condition during the filling operation of cosmetics and pharmaceuticals. According to the results of this investigation,
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)