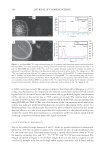
J. Cosmet. Sci., 60, 315–321 (May/June 2009) 315 In vivo skin analysis (INSA) for quantitative determination of lotion transfer to human skin ARMAN EBRAHIMPOUR and ALAN H. ULLMAN, Procter & Gamble Co., 8700 Mason-Montgomery Road, Cincinnati, OH 45040 (A.E.), and Procter & Gamble Co., 6100 Center Hill Avenue, Cincinnati, OH 45224 (A.H.U.). Accepted for publication January 9, 2009. Synopsis There is a need during the development of cosmetic and skin products for simple, quantitative, noninvasive measurements of product deposition onto skin. In this article we describe INSA (in vivo skin analysis) as such a method for measuring the amount of lotion transferred to the skin from tissue products. Using Fourier transform infrared spectroscopy with an attenuated total refl ectance (ATR FT-IR) sampling accessory, we were able to quantify lotion levels on the arms of subjects in minutes. INTRODUCTION The measurement of materials applied to skin is important to both the cosmetic and health care industries. Of the measurement approaches in the literature, one of the most common ones involves literally removing layers of skin from the subject (by tape stripping) for analysis. Typically, this tape stripping is performed on the subject, and the combined tape and skin layers are extracted with solvent for analysis by spectroscopic techniques or by gas or liquid chromatography (1–3). This method is invasive and relatively slow extraction times in excess of 30 minutes and chromatograph run times of an additional 30 minutes are common. In addition, tape stripping and extraction effi ciency—for all compounds of interest—must be established. Furthermore, due to its invasive nature, panelists subjected to tape stripping are not eligible for repeat testing (on the affected area) for several weeks. However, where depth of penetration of a compound into the skin is studied, it is the method of choice. In recent years, the use of paper and nonwoven substrates, such as wipes and tissues, to deliver lotions and actives to the skin has gained wide prominence. Thus, measurement of the amount of lotion transferred from the tissue to the skin is needed to assist in the development of improved products. Attenuated total refl ectance Fourier transform infrared spectroscopy (ATR FT-IR) is a well-known technique for the measurement of chemicals on surfaces (4,5). Using this technique, penetration of the evanescent IR wave into the surface is on the order of
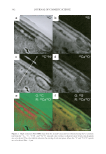
JOURNAL OF COSMETIC SCIENCE 316 micrometers, depending primarily on the refractive index of the ATR crystal. For exam- ple, for a zinc selenide (ZnSe) crystal at 1000 cm−1 and a sample with a refractive index of 1.5 and an angle of incidence of 45°, the penetration is about 2 μm. As a nondestructive, noninvasive technique, it is ideal for skin measurements and has been used for such measurements in the past (6). However, direct calibration of the technique has proven diffi cult. Application of quantitative standards to the skin has been used (6), but it may not be safe for low levels of analyte because of the solvents needed to dilute the analyte. In addition, the solvents may change the extent of absorption of the applied compounds. Direct application to the ATR crystal of analytes in dilute solution, however, is a tech- nique that yields excellent results. EXPERIMENTAL APPARATUS A BioRad model 575C enhanced intensity FT-IR spectrometer with Pike Technologies’ ATR skin analyzer accessory was used in this work. The ATR crystal was made of zinc selenide with a fl at geometry of 45°, and was 0.40-cm thick and 1.0-cm wide by 10.0-cm long. DigiLab Win-IR Pro software was used to collect the spectra on the PC platform computer. The ATR-FT-IR instrument was aligned prior to the experiments, and all spectra were collected between 4000 and 700 cm−1. Hamilton Gastight® syringes (0.500 ml, #1750) were used to assure accurate delivery of solvents and solutions. REAGENTS Hexane Spectranal® grade (Riedel-de Haën from Sigma Aldrich) was used as solvent in preparing all standards and for rinsing the ATR crystal, glassware, etc. High-purity sol- vent was found to be critical to the method (see below.) PROCEDURES In order to calibrate the instrument, a spectrum of a known amount of analyte must be measured while the skin is in contact with the ATR crystal. Ideally, the standard would be applied to the subject’s arm with the known amount of analyte dissolved in solvent, but solvent interaction with the skin would likely change the skin lipids, the penetration of analyte into the skin, or any number of other important parameters. Depositing the calibration standards directly onto the crystal in dilute solution eliminated these issues. Known volumes of known concentration solutions were carefully delivered by calibrated Gastight syringes, taking care to distribute the solution across the surface of the crystal. The choice of solvent was critical: while solvent must evaporate from the crystal, it must not evaporate too rapidly since the solution may not be delivered quantitatively to the surface. For example, dichloromethane did not pass this requirement. Furthermore, the solvent must be one that evaporates fully from the crystal, leaving no residue. Only high- purity solvents are acceptable early attempts with regular laboratory grade hexane left a residue that produced absorption peaks in the same area, 3200–2600 cm−1 (the C–H
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)