LOTION TRANSFER TO SKIN 317 region), as the lotion we were measuring. Most lotion substrate products, including lotioned facial tissues, have as primary lotion components petrolatum and mineral oil, with key absorbance peaks in this region. Calibration method. The ATR FT-IR apparatus was calibrated for spectral response versus the amount of lotion deposited onto the ATR crystal, with and without the presence of hu- man skin on the crystal. The lotion used for calibration and analysis was similar to those used most commonly in consumer substrate products such as facial tissues. The main com- ponents in these lotions were mineral oil, petroleum wax, cetyl, and stearyl alcohols. A standard stock solution of the lotion in hexane was prepared by weight at 5071.97 μg lotion per gram of solution. From the standard stock solution ten calibration standards were pre- pared by dilution with hexane, ranging from 9 to 1230 μg/g. The spectra of each standard solution were obtained, with and without skin contact, using 0.250 ml of the given stan- dard solution, spread over the entire surface (10 cm2) of the ATR crystal. The human skin portion used in these studies was on the subjects’ volar forearms subjects’ selection criteria, prerequisites, and skin preparation methods are detailed in the next section. Human skin study design. The following study protocol was adhered to throughout this study. Participation in the study was completely voluntary. At the initial visit, subjects were screened for any conditions that would exclude them from the study. Subjects were screened for study eligibility based on the following criteria. Excluded from the study was anyone who: (a) was participating in another clinical trial (b) had an excess amount of hair on the volar forearm (c) was perspiring excessively due to laborious work or exercise, thus resulting in increased skin moisture (d) had allergies to soap, detergent, perfume, cosmetics, toiletries, and/or lotioned tissue products (e) had eczema or psoriasis on the forearms within the past six months (f) had been diagnosed with skin cancer within the past twelve months (g) had any cuts, scratches, rashes or any condition on their inner forearms (e.g., damaged skin, extensive scarring, birth marks, port wine stains, tattoos, moles) that may pre- vent a clear assessment of their skin (h) was using or had used, within the past two weeks, any topical prescription or over- the-counter (OTC) medications (e.g., hydrocortisone cream, antibiotic ointment) on their forearms (i) was a smoker The entire volar forearm was submerged in running water (37°C ± 5°C), washed with a liquid detergent (e.g., 2 ml Dawn® dishwashing detergent), rinsed with running water until all of the soapy water was removed, and then blotted dry with a lotion-free paper towel (e.g., Bounty® paper towel). Subsequently, a timer was set and the forearm was al- lowed to dry for at least two minutes. After drying, the washed area of the volar forearm was positioned on the ATR prism and the FT-IR spectrum was collected. While the spec- trum was being acquired, the subject’s arm remained motionless. The scan was completed within one minute. Prior to each measurement, the prism was cleaned with Kimwipes® and solvent. A blank spectrum was collected to be certain the crystal was clean. This initial scan on cleaned skin became the baseline scan of the volar forearm. Use of this clean arm as “control” helped negate differences in arm pressure on the ATR surface.
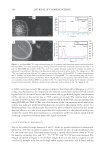
JOURNAL OF COSMETIC SCIENCE 318 Subsequently, the test product was wiped across the subject’s entire volar forearm. Prod- ucts were commercially available facial tissue products with dimensions of about 21 cm by 21 cm (441 cm2). Tissues were folded so that an area of about 25 cm2 was wiped across the forearm. The same volar area of the forearm was then placed on the prism area of the ATR and scanned. (As the lotion contained no water or other solvent, no “drying” time was needed between the tissue wipe on the arm and placement of the arm on the ATR crystal.) The same procedure was repeated on the other forearm. The forearms were alternated between test products used. Application of the product onto the volar forearm used sim- ilar force by the same study personnel. RESULTS AND DISCUSSION CALIBRATION METHOD All ATR FT-IR spectra were baseline corrected, and the maximum peak height at 2914 cm−1 recorded. Figure 1 shows spectra of clean skin and skin+lotion on the ATR prism. Table I summarizes the lotion deposition vs ATR FT-IR spectra results used to derive the calibration line equation for the INSA method. These values are measured at the maximum baseline- corrected spectral peak height at 2914 cm−1. These results are also illustrated in Figure 2, where the solid line represents the spectral response in the absence of skin and the hatched line represents the spectral response while the prism is in close contact with the forearm skin. The presence or absence of skin against the ATR prism did not play an important role in the overall spectral response to the lotion deposition on the prism the slopes and intercepts of the regression lines were nearly identical. Furthermore, the responses in both cases were Figure 1. INSA spectra of cleaned skin before (dashed line) and after (solid line) lotion product application.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)