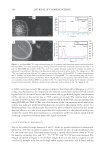
JOURNAL OF COSMETIC SCIENCE 336 The authors owe thanks to Dr. Steven Breakspear of Kao Corporation for his valuable comments and advise on this paper. The authors are also obliged to Ms. Masae Sakaguchi for taking the beautiful fi ber picture of total refl ection. REFERENCES (1) P. Henry, Br. J. Appl. Phys., 4, 531 (1953). (2) T. L. Ward and R. R. Benerito, Correlation of visual luster with measured refl ectance of cotton fabrics, Textile Res. J., 35, 271 (1965). (3) L. Fourt, Improvement of luster of wool fabrics, Textile Res. J., 36, 915 (1966). (4) W. Thompson and C. Mills, Proc. Sci, Sect. T.G.A., No. 15 (1951). (5) R. F. Stamm, M. L. Garcia, and J. J. Fuchs, The optical properties of human hair. I. Fundamental con- siderations and goniophotometer curves, J. Soc. Cosmet. Chem., 28, 571 (1977). (6) R. F. Stamm, M. L. Garcia, and J. J. Fuchs, The optical properties of human hair. II. The luster of hair fi ber, J. Soc. Cosmet. Chem., 28, 601 (1977). (7) W. J. Conover, Practical Nonparametric Statistics (John Wiley & Sons, New York, 1980), p. 299. (8) J. H. S. Rennie, S. E. Bedford, and J. D. Hague, A model for the shine of hair arrays, Int. J. Cosmet. Sci., 19, 131 (1997). (9) C. Reich and C. R. Robbins, Light scattering and shine measurements of human hair: A sensitive probe of the hair surface, J. Soc. Cosmet. Chem., 44, 221 (1993). (10) R. Meredith and J. W. S. Hearle, Physical Methods of Investigating Textiles (Interscience, New York, 1959), Ch. 12. (11) H. K. Bustard and R. W. Smith, Investigation into the scattering of light by human hair, Appl. Optics, 30, 3485 (1991). (12) W. Czepluch, G. Hohm, and K. Tolkiehn, Gloss of hair surfaces: Problems of visual evaluation and pos- sibilities for goniophotometric measurements of treated strands, J. Soc. Cosmet. Chem., 44, 299 (1993). (13) N. Shinobu, S. Shibuichi, A. Kenichi, K. Emiko, and S. Naoki, Infl uence of internal structure of hair fi ber on hair appearance. I. Light scattering from the porous structure of the medulla of human hair, J. Cosmet. Sci., 53, 89 (2002). (14) N. Shinobu, S. Naoki, and N. Koichi, Infl uence of internal structure of hair fi ber on hair appearance. II. Consideration of the visual perception mechanism of hair appearance, J. Cosmet. Sci., 53, 387 (2002). (15) O. Masayuki, Y. Ryoko, M. Akira, I. Shigeto, N. Shinobu, S. Shibuichi, K. Emiko, and S. Naoki, Infl u- ence of internal structure of hair fi ber on hair appearance. III. Generation of light-scattering factors in hair cuticles and the infl uence on hair shine, J. Cosmet. Sci., 54, 353 (2003). (16) S. Satoshi, S. Hirayuki, W. Shunsuke, K. Tetsuya, and S. Naoki, Mechanism of blonde hair appearance, 14th International Hair-Science Symposium, ‘HairS’ 05, 2005. (17) A. Franbourg, P. Hallegot, F. Baltenneck, C. Toutain, and F. Leroy, Current research on ethnic hair, J. Am. Acad. Dermatol., 48, S115 (2003).
J. Cosmet. Sci., 60, 337–345 (May/June 2009) 337 Copper and calcium uptake in colored hair K. E. SMART, M. KILBURN, M. SCHROEDER, B. G. H. MARTIN, C. HAWES, J. M. MARSH, and C. R. M. GROVENOR, Department of Materials, University of Oxford, Parks Road, Oxford, OX1 3PH, UK (K.E.S, M.S, C.R.M.G.), School of Life Sciences, Oxford Brookes University, Gipsy Lane, Oxford, OX3 0BP, UK (B.G.H.M., C.H.), Procter & Gamble, Miami Valley Innovation Center, 11810 E. Miami River Road, Cincinnati, OH 45252 (J.M.M.), and Centre for Microscopy, Characterisation & Microanalysis, University of Western Australia, 35 Stirling Highway, Crawley, 6009 WA, Australia (M.K.). Accepted for publication December 29, 2008. Synopsis During hair coloring a number of disulfi de bonds in cystine are oxidized (1) to create cysteic acid, forming binding sites for metal ions such as Ca2+and Cu2+from tap water (2). The increased uptake of these metals can have a detrimental impact on fi ber properties—for example, reducing shine and causing a poor wet and dry feel (3). In addition, the increased uptake of copper can also contribute to further fi ber damage during subse- quent coloring due to its ability to take part in metal-induced radical chemistry (4). It is important to know where in the fi bers these metals are located in order to either effectively remove these metals or control their chemistry. Nanoscale secondary ion mass spectrometry (NanoSIMS) has been used to locate the calcium and copper within hair that has been treated with a colorant and washed multiple times in tap water containing these ions. Untreated hair is used as a baseline standard material. Images with up to 50-nm spatial resolution of the preferential locations of calcium uptake were obtained, showing a high concentration of calcium in the cuticle region of colored hair, specifi cally in the sulfur-rich regions (A-layer and exocuticle). INTRODUCTION Typical permanent hair colorants contain hydrogen peroxide buffered to a pH of 10 with ammonium hydroxide. The role of the oxidant is to bleach the melanin, lightening the under- lying substrate color, and to oxidize the dye precursors to form chromophores. The fi nal color the consumer achieves is a combination of lightening of the natural color and deposi- tion of synthetic color inside the hair. However, the oxidant can also react with the hair proteins and lipids, leading to changes in the fi ber properties that can be experienced by consumers, especially over multiple cycles. These properties include a reduction in shine, reduced manageability, poor wet and dry feel, and increased likelihood of split ends (3). One of the proposed mechanisms of fi ber damage is reaction of the hair proteins and lipids with hydroxyl radical species that are formed from the catalytic reaction between hydrogen Address all correspondence to J. M. Marsh.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)