JOURNAL OF COSMETIC SCIENCE 338 peroxide and redox metals such as copper (4). The source of the copper is the tap water used during the shampoo and rinsing processes (5). Water hardness ions such as calcium and magnesium are also taken up by hair from the tap water. Although the calcium does not directly take part in the color chemistry, it can affect the shine and combing properties of the hair by forming insoluble calcium salts and soaps on the fi ber surface (6). For both met- als it has been demonstrated that their uptake increases as the hair undergoes multiple treat- ments from an oxidative colorant. It has been hypothesized that this is due to the enhanced number of metal binding sites that are formed in the hair during the oxidation process. The presence of metals in hair has been widely reported in the literature, with earlier work concentrating on measuring the range and levels of metals present in the fi ber (7). This work also demonstrated that there are two main sources of these metals, endogenous and exogenous. The endogenous metals are incorporated in the fi ber at the hair follicle and are thought to act as a pathway for the body to excrete unwanted metals (8). The exogenous metals come from the fi ber’s exposure to the environment and in particular to the water used in its washing (9). Recent research has focused on identifying the specifi c location of metals in the fi ber structure and on identifying which metals are exogenous and which metals are endogenous. Of particular interest has been the presence of metals whose origin may have been due to exposure to pollution. Kempson and Skinner (10) investigated the hair from a worker exposed to lead, and Audinot et al. (11) investigated hair from an individual exposed to arsenic. In both cases the nature of the metal as either exogenous or endogenous was identifi ed. ToF SIMS and x-ray microfl uorescence imaging have been used to map calcium in hair and to identify the source as either exogenous or endogenous (12,13). The technique used here to investigate the location of metals is nanoscale secondary ion mass spectrometry (NanoSIMS). SIMS is a surface analysis technique in which a focused primary ion beam is scanned across the target surface. The bombardment of these primary ions results in the ejection of charged atomic and molecular species from the sample sur- face. The secondary ions are then separated on the basis of their mass-to-charge ratio and correlated to their spatial origin to form a chemical image (14,15). The NanoSIMS is the latest generation of dynamic SIMS facilities and is capable of achieving, at the same time, extremely high spatial resolution ( 50 nm) and mass resolving power (m/∆m = 5,000) due to its coaxial lens confi guration and advanced ion optics. Limited previous work has been done using this kind of analysis on hair samples. The work of Hallegot and Corcuff (16) was the fi rst example of a SIMS study of a hair fi ber, while Collin et al. (17) studied the presence of isotopically labeled taurine in hair. In this study we have concentrated on studying the location of both calcium and copper in hair samples, both untreated and after treatment with an oxidative colorant. EXPERIMENTAL SAMPLE PREPARATION Three samples were used for analysis. The fi rst was untreated Caucasian hair, the second was colored Caucasian hair, and the third was colored Caucasian hair containing high levels of copper. The Caucasian hair containing high levels of copper was used as a control to check the sensitivity of the instrument to detect copper in the hair samples.
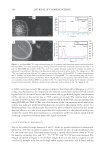
Cu AND Ca UPTAKE IN COLORED HAIR 339 Caucasian untreated mixed hair (natural white), obtained from a commercial source (IHIP, New York), was formed into swatches (16 cm, 1.5 g). The treated hair was obtained by treating the untreated hair with a commercial extra-light blonde shade that contained 4.5% hydrogen peroxide and ammonium hydroxide at a pH of 10. Four grams of the product was applied to each gram of hair. The product-covered hair was placed in an oven for 30 minutes at 30°C and then rinsed for 1 min in tap water containing about 300 ppm calcium and magnesium, and approximately 0.06 ppm copper. The hair was then washed twelve times using 0.1 g of a commercial shampoo. This whole process, involving one color and twelve washes, was repeated fi ve times to create the treated Caucasian white hair sample. The color- treated hair was placed under running tap water dosed with copper chloride (1 ppm) for 30 minutes to create the copper-rich sample. The hair was then dried in front of a fan. To create a fl at surface for NanoSIMS analysis, the hair was encapsulated within a resin matrix. The hair was cut into 2-cm lengths, placed in a shallow aluminium dish contain- ing medium-grade L.R. White resin, and fully coated in resin. Small bundles of resin- coated hair were positioned vertically in 1.5-ml Eppendorf tubes and polymerized in the oven at 70°C for 9 hr. The hair-containing resin blocks were then cut into sections of 0.5-μm thickness using a RMC powertomeXL ultramicrotome. The thin sections were placed on a droplet of water on a silicon substrate and warmed gently to give good adher- ence. All samples were gold coated to maintain conductivity in the NanoSIMS. HIGH-RESOLUTION SECONDARY ION MASS SPECTROMETRY The CAMECA NanoSIMS 50 can be operated with a Cs+primary beam to detect negative secondary ions, or with an O− primary beam to detect positive secondary ions. High- resolution ion images were obtained using the Cs+ primary ion beam, focused to approxi- mately 50 nm. The ion species 12 C−, 32 S−, 12 C14N−, and 40 Ca16O− were simultaneously mapped over an area of 5 × 5 μm, with 512 × 512 pixels in each image. The O− primary beam, with a resolution of about 300 nm, was used to map 40 Ca+ and 63 Cu+ over an area of 40 × 40 μm, with 256 × 256 pixels in each image. Each area was cleaned and implanted with the primary beam prior to image acquisition. To improve the counting statistics, fi ve sequential images were added together. INDUCTIVELY COUPLED MASS SPECTROMETRY Samples (0.5 g) were taken from each of the three types of hair, washed, and then analyzed using inductively coupled plasma-mass spectroscopy (ICP-MS). RESULTS AND DISCUSSION CALCIUM MAPPING: O- BEAM RESULTS In the fi rst set of experiments we investigated the use of the O− beam for chemical map- ping of both the uncolored and colored hair samples. The 40 Ca+ and 63 Cu+ images are shown respectively in Figure 1(a,b) for untreated hair and in Figure 1(d,e) for treated hair. The untreated hair shows an enhanced concentration of calcium in the cuticle region (~1000 counts per second) and also at the center of the fi ber, in the medulla region (2000
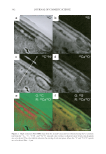
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)