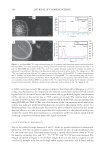
STABILITY OF 8-HYDROXYDAIDZEIN 355 Figure 2. Degradation of 8-hydroxydaidzein in a series of buffers with different pH values. The buffers used were 50 mM acetate buffer for pH 5 (♦) and pH 6 (■) 50 mM phosphate buffer for pH 7 (▲) and 50 mM Tris-HCl buffer for pH 8 (—) and pH9 (●). The experimental conditions were the same as those in Figure 1. the compound in the pH 6.8 phosphate buffer. One is that the compound might react with phosphate and result in instability. Another reason is that the compound was actu- ally not soluble in the tested water system and cannot be detected by the HPLC method. The last one is that the pH of the solvent could affect the stability of the compound. The fi rst two reasons were quickly ruled out because similar instability phenomena of the compound were also found when the phosphate solvent was replaced by 50 mM of pH 6.8 Tris-HCl buffer and the 8-hydroxydaidzein solutions prepared by either phosphate or Tris-HCl buffer were not found to produce any precipitation after centrifugation with 12,000 rpm for 10 min. Hence, the most likely factor that caused the instability of 8-hydroxydaidzein in the solution is the pH of the solvent used. To prove the point, ex- periments similar to those described above were repeated, but using buffers with different pH values to prepare the 8-hydroxydaidzein solutions. The tested buffers included 50 mM acetate buffer for pH 5 and pH 6, 50 mM phosphate buffer for pH 7, and 50 mM Tris-HCl buffer for pH 8 and pH 9. The stability of 8-hydroxydaidzein in different solutions was analyzed by the HPLC method. The results are shown in Figure 2. When the compound was dissolved in Tris-HCl buffers of pH 8 and pH 9, it was quickly degraded within 10 hours. In contrast, the compound remained stable above 85% after 20 days’ storage in acetate buffers of pH 5 and pH 6. In addition to the HPLC analysis of residual 8-hydroxydaidzein, the residual bioactivities of the 8-hydroxydaidzein solutions with different pH values were also determined at the end of the storage experiment. As shown in Figure 3, tyrosinase inhibitory activity and DPPH-radical scavenging activity of the solutions remained at 94% and 82% in the pH 5 acetate buffer and at 93% and 78% in the pH 6 acetate buffer, respectively, while no activity could be detected in the 8-hydroxydaidzein solutions prepared in the pH 8 and pH 9 Tris-HCl buffers. The results of both residual tyrosinase inhibitory activity and DPPH-radical scavenging activity of the prepared 8-hydroxydaidzein solutions with different pH values correlate well with those of residual 8-hydroxydaidzein in each solution. The results of the present study demonstrate that 8-hydroxydaidzein is unstable in alkaline solutions. CONCLUSIONS Thermal stability of soy isofl avones has been studied well in soy fl our (5), soy milk (6), and model solutions (7). However, little has been reported about the effects of pH on the
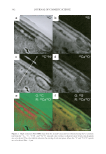
JOURNAL OF COSMETIC SCIENCE 356 stability of soy isofl avones. In this study, a soy isofl avone derivative, 8-hydroxydaidzein, was demonstrated to be unstable in alkaline solutions. In fact, daidzein itself, the bio- transformed precursor of 8-hydroxydaidzein, would be stable in both pH 7 and pH 9 buffers at 25°C (data not shown). Hence, to understand the detailed mechanism causing the instability of 8-hydroxydaidzein in alkaline solutions, more study is needed. Besides being a potent irreversible inhibitor toward mushroom tyrosinase (3–4), 8- hydroxydaidzein also contains strong antioxidant activity (1–2), anti-mutagenic activity (8), anti-aldose reductase activity (9), and anti-proliferation activity toward cancer cell lines (10). Because of the multifunctional bioactivities of 8-hydroxydaidzein, it is ex- pected that broad application and high quality will be needed for storage of the com- pound. Hence, according to the data in the present report, it is recommended that 8-hydroxydaidzein be formulated in an acid solution for its application in whitening cosmetics, foods, or medical agents. REFERENCES (1) H. Esaki, H. Onozaki, Y. Morimitsu, S. Kawakishi, and T. Osawa, Potent antioxidant isofl avones iso- lated from soybeans fermented with Aspergillus saitoi, Biosci. Biotechnol. Biochem., 62, 740–746 (1998). (2) H. Esaki, R. Watanabe, H. Onozaki, and S. Kawakishi, Formation mechanism for potent antioxidative 0-dihydroxyisofl avones in soybeans fermented with Aspergillus saitoi, Biosci. Biotechnol. Biochem., 63, 851–858 (1999). (3) T. S. Chang, H. Y. Ding, S. S. K. Tai, and C. Y. Wu, Tyrosinase inhibitors isolated from soygerm koji fermented with Aspergillus oryzae BCRC 32288, Food Chem., 105, 1430–1438 (2007). (4) T. S. Chang, Two potent suicide substrates of mushroom tyrosinase: 7,8,4′,-trihydroxyisofl avone and 5,7,8,4′,-tetrahydroxyisofl avone, J. Agric. Food Chem., 55, 2010–2015 (2007). (5) Y. Chen, M. Inaba, N. Abe, and A. Hirota, Antimutagenic activity of 8-hydroxyisofl avones and 6- hydroxydaidzein from soybean miso, Biosci. Biotechnol. Biochem., 67, 903–906 (2003). Figure 3. Residual tyrosinase inhibitory activity (vertical lines) and DPPH-radical scavenging activity (hor- izontal lines) of 8-hydroxydaidzein in a series of solutions with different pH values. For the tyrosinase in- hibitory activity analysis, 100 μl of the tested sample was pre-incubated with 400 μl of tyrosinase (20 units) in 50 mM of phosphate buffer (pH 6.8) at 25°C for 30 min. Then, 500 μl of 2.5 mM L-Dopa in the same phosphate buffer was added and the reaction mixture was incubated at 25°C for 10 min. The formation of dopachrome in each reaction was monitored at 475 nm with a spectrophotometer. For the DPPH-radical scavenging activity analysis, 100 μl of the tested sample was mixed with 900 μl of 2 mM DPPH MeOH solu- tion. After standing for 30 min, the absorbance of the mixture at 517 nm was measured. The tested samples included a freshly prepared 8-hydroxydaidzein solution [100 μg/ml in 50 mM of phosphate buffer (pH 6.8)] and the prepared solutions at the end of the stability experiment in Figure 2. The relative activity was calcu- lated by dividing the absorbance of each reaction mixture by that of the control reaction, in which phosphate buffer replaced the tested compound. The residual activity of each sample was calculated by dividing the relative activity of each sample by that of the freshly prepared sample.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)