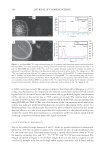
TYROSINASE INHIBITORS FROM P. SUFFRUTICOSA 351 this study. One tannin compound pentagalloyl-β-D-glucose (VI) exhibited the most po- tent inhibitory activity toward mushroom tyrosinase among the six inhibitors purifi ed from the root of P. suffruticosa. Its inhibitory activity (IC50 value 0.063 mM) was slightly higher than that of kojic acid (IC50 value 0.070 mM). In addition, the compound’s inhi- bition kinetics was analyzed by the Lineweaver-Burk plot method, and the results are shown in Figure 2. The three lines, obtained from the uninhibited enzyme and the two concentrations of the inhibitor, intersect on the horizontal axis. These results demonstrate that pentagalloyl-β-D-glucose (VI) exhibited non-competitive inhibition of the oxidation of L-Dopa catalyzed by mushroom tyrosinase. P. suffruticosa is an important Chinese traditional medicine and acts as an antibiotic and a hemorrhage inhibitor. The present study identifi ed six tyrosinase inhibitors from the plant’s roots. Among them, the newly identifi ed tyrosinase inhibitor, pentagalloyl-β-D-glucose (VI), is a potent non-competitive inhibitor and shows slightly higher activity than that of kojic acid. In this study, kojic acid was used as a reference standard of tyrosinase in- hibitor due to the in vitro assay model used. Besides kojic acid, hydroquinone is known as an effective inhibitor of melanogenesis. However, hydroquinone is rarely used as a refer- ence standard in the in vitro tyrosinase inhibitory assay due to its lower tyrosinase inhibi- tory activity than that of kojic acid (2). The potent inhibitory activity on melanogenesis of hydroquinone is based on its multifunctional effects. Besides tyrosinase inhibitory activity, hydroquinone possesses many other activities in melanogenesis inhibition, such as alteration of melanosome functions, depletion of glutathione, generation of reactive Table I Inhibitory Effects of Tyrosinase Inhibitors Isolated from Root of P. suffruticosa on Mushroom Tyrosinase Activity (Substrate: L-Dopa) Sample Inhibitory activity (IC50 mM) Inhibition mode Kaempferol (I) 0.120 ± 0.006 Competitive Quercetin (II) 0.108 ± 0.009 Competitive Mudanpioside B (III) 0.368 ± 0.008 Competitive Benzoyloxypaeonifl orin (IV) 0.453 ± 0.012 Competitive Mudanpioside H (V) 0.324 ± 0.026 Competitive Pentagalloyl-β-D-glucose (VI) 0.063 ± 0.001 Noncompetitive Kojic acid 0.070 ± 0.001 Mixeda a Obtained from data in reference 18. Figure 2. Lineweaver-Burk plots of mushroom tyrosinase and L-Dopa without (♦) and with 17.5 (▲) and 35 (■) μg/ml pentagalloyl-β-D-glucose (VI).
JOURNAL OF COSMETIC SCIENCE 352 oxygen species, and subsequent oxidative stress-mediated melanocytotoxic effect. Hence, just like kojic acid in tyrosinase inhibitors, hydroquinone is an excellent standard in evaluating depigmenting agents. However, because of the melanocytotoxic effect, hydro- quinone is hazardous in long-term treatments, and the use of hydroquinone in cosmetics has been limited (17). According to the above description, comparing both the melano- genesis inhibitory activity and the melanocytotoxic effect of pentagalloyl-β-D-glucose (VI) with the activities of hydroquinone in the future would provide clearer evidence for its application in skin-whitening cosmetics. REFERENCES (1) S. Y. Seo, V. K. Sharma, and N. Sharma, Mushroom tyrosinase: Recent prospects, J. Agric. Food Chem., 51, 2837–2853 (2003). (2) S. Briganti, E. Camera, and M. Picardo, Chemical and instrumental approaches to treat hyperpigmenta- tion, Pig. Cell Res., 16, 101–110 (2003). (3) Y. Masamoto, H. Ando, Y. Murata, Y. Shimoishi, M. Tada, and K. Takahata, Mushroom tyrosinase in- hibitory activity of Esculetin isolated from seeds of Euphorbia lathyris L., Biosci. Biotechnol. Biochem., 67, 631–634 (2003). (4) T. S. Chang, H. Y. Ding, and H. C. Lin, Identifying 6,7,4′-trihydroxyisofl avone as a potent tyrosinase inhibitor, Biosci. Biotechnol. Biochem., 69, 1999–2001 (2005). (5) S. Arichi, M. Kubo, H. Matsuda, T. Tani, K. Tsunaga, M. Yoshikawa, and I. Kitagawa, Studies on mou- tan cortex (III). On anti-infl ammatory activities (Part 1), Shoyakugaku Zasshi, 33, 178–184 (1979). (In Japanese) (6) C. Chen, Research and Development of the Cosmetic Health Products of Traditional Chinese Medicine (Chinese Medica Technological Publishing Co., Beijing, 1998), pp. 453–456 (in Chinese). (7) M. Yoshikawa, E. Uchida, A. Kawaguchi, I. Kitagawa, and J. Yamahara, Galloyl-oxypaeonifl orin, suf- fruticosides A, B, C, and D, fi ve new antioxidative glycosides, and suffruticoside E, a paeonol glycoside, from Chinese moutan cortex, Chem. Pharm. Bull., 40, 2248–2250 (1992). (8) M. Yoshikawa, E. Uchida, A. Kawaguchi, J. Yamahara, N. Murakami, and I. Kitagawa, Absolute stereostructures of paeoniffrone and paeonisuffral, two new labile monoterpenes, from Chinese moutan cortex, Chem. Pharm. Bull., 41, 630–632 (1993). (9) H. C. Lin, H. Y. Ding, T. S. Wu, and P. L. Wu, Monoterpene glycosides from Paeonia suffrutticosa, Phytochem., 41, 237–242 (1996). (10) H. C. Lin, H. Y. Ding, and Y. C. Wu, Two novel compounds from Paeonia suffrutticosa, J. Nat. Prod., 61, 343–346 (1998). (11) H. Y. Ding, Y. C. Wu, H. C. Lin, Y. Y. Chan, P. L. Wu, and T. S. Wu, Glycosides from Paeonia suffrut- ticosa, Chem. Pharm. Bull., 47, 652–655 (1999). (12) H. Y. Ding, H. C. Lin, C. M. Teng, and Y. C. Wu, Phytochemical and Pharmacological studies on Chi- nese Paeonia species, J. Chin. Chem. Soc., 47, 381–388 (2000). (13) I. Kubo, I. Kinst-Hori, S. K. Chaudhuri, Y. Kubo, Y. Sanchez, and T. Ogura, Flavonols from Heterotheca inuloides: Tyrosinase inhibitory activity and structural criteria, Bioorg. Med. Chem., 8, 1749–1755 (2000). (14) H. Matsuda, M. Higashino, W. Chen, H. Tosa, M. Iinuma, and M. Kubo, Studies of cuticle drugs from natural sources. III. Inhibitory effect of Myrica rubra on melanin biosynthesis, Biol. Pharm. Bull., 18, 1148–1150 (1995). (15) C. H. Jeong and K. H. Shim, Tyrosinase inhibitor isolated from the leaves of Zanthoxylum piperitum, Biosci. Biotechnol. Biochem., 68, 1984–1987 (2004). (16) I. Kubo and I. Kinst-Hori, Flavonols from saffron fl ower: Tyrosinase inhibitory activity and inhibition mechanism, J. Agric. Food Chem., 47, 4121–4125 (1999). (17) A. P. De Caprio, The toxicology of hydroquinone-relevance to occupational and environmental expo- sure, Crit. Rev. Toxicol., 29, 283–330 (1999). (18) J. S. Chen, C. Wei, R. S. Rolle, W. S. Otwell, M. O. Balaban, and M. R. Marshall, Inhibitory effect of kojic acid on some plant and crustacean polyphenol oxidases, J. Agric. Food Chem., 39, 1396–1401 (1991).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)