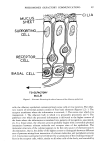
PHEROMONES (OLFACTORY COMMUNICATION) 55 old subjects showed essentially no response. At two weeks they exhibited general arousal with little attendant discrimination between their mother's pad and those of strange mothers. At six weeks of age, six of the ten infants tested could identify the ma- ternal odor from that of a stranger. In an earlier and independent investigation (57), a differential response was shown six days after birth. The source of these odors is not known with certainty. The response may be due to a true maternal odor or, as demonstrated in other primates (58), originate in odors placed on the mother by the neonate. In either instance, olfactory cues generate behavioral responses in infants. The ontogenetic implications are fascinating. For example, would the putative maternal odor or similar odor "imprinted" between one and six weeks give rise to a behavioral response later in the adult? Regarding the effects of odor on developing Homo sapiens, Bieber (59) stated, "The sense of smell is the primary sensory modality in the heterosexual development of heterosexual responsivity." Furthermore, it has been suggested (60) that olfaction may play a crucial role in the establishment of sexual identity. An interesting study (61) has demonstrated that adult male hamsters undergo a dra- matic change toward the female's vaginal secretion when its presentation was followed by lithium chloride poisoning. The secretion, which is sniffed and ingested by the male, contains a substance that specifically affects sexual motivation which does not depend on previous experience. Surprisingly, hamsters form aversions to the vaginal secretion when it has been followed by a single experience with poisoning. The experiments sug- gest that adult mammals' responses to sex pheromones are much more easily modified by experience than has been supposed. The olfactory system, in consort with limbic structures, served to free animal behavior from the rigidity of inborn reflex mechanisms mediated by the hypothalamus, e.g., temperature regulation and the au- tonomic control of cardiovascular and digestive systems. As a result, not only are specific, genetically programmed pheromonal effects made possible, but also those associated with past life experiences. The sexual behaviour of the male rhesus monkey to an odorant emitted by the female tended to support the belief held by some scientists and laymen (62-64) that specific, naturally occurring body odors ("pheromones") may have previously unsuspected influences on social and sexual behaviors of man. Michael and Keverne (ll-14) reported that male rhesus monkeys are strongly attracted to and copulated with estrogen-treated females, presumably on the basis of olfactory cues. A series of low molecular weight aliphatic acids, i.e., acetic, propionic, isobutyric, butyric and iso- valeric, were proposed to be the active components of the vaginal secretions (15). The acid mixture, when substituted for the vaginal secretions, was said to have similar stimulatory effects on the males. In view of the basic importance of these findings to a general theory of primate behavior, Goldfoot and co-workers (17,18) performed addi- tional evaluations of the effects of vaginal material on the male rhesus sexual behavior. Their detailed st•udy failed to support the conclusions drawn from previously published work, that is, vaginal secretions and aliphatic acids essentially were without effect on the males. The significant procedural differences between the two groups of investiga- tors lie in Michael et al's practice of prior selection of males and on specific experiences provided them which, in effect, trained the males to respond to the odor cues (18). Al- though a clearer picture will emerge after further experimentation, the effect of associative learning in the rhesus monkey rather than a pheromonic phenomenon ap- pears to best explain the evidence accumulated to date.
56 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Failure of an animal to subsequently respond to an arbitrarily selected odorant used in a learning experiment does not necessarily signify the lack of capability for olfactory communication. Seligman and Hager (65) have shown that learning has its biological boundaries. They make a most persuasive case for the "preparedness" (66-68) hypothesis in behavior studies in contrast to that of"equipotentiality" (69,70). In the latter, it is assumed that all perceptible stimuli are equivalent. Implicit in the prepared- ness hypothesis is the premise that learning is continuous with instinct. Lockhard (71) wrote, ".... natural selection has produced special learning abilities such that some ecologically relevant task is learned at a much faster rate than an arbiyrary task, or natural stimuli are much more effective than artificial stimuli." It follows that the chances of a successful associative learning experiment would be increased when a bio- logically significant odor cue is used. Although there have been conjectures (62,63) concerning apocrine gland, skin and vaginal secretions serving as olfactory communi- cants, little is known of the effects of these odors on man. Behavioral responses elicited by olfactory messengers probably are strongly influenced by the context within which odorants are presented. Also, human volatile secretions may vary with emotional state. More than 70 years ago Ellis (72) wrote, "Women, like men, frequently give out an odor during coitus or strong sexual excitement. This odor may be entirely different from that normally emanating from the woman." Another comment of this prescient psychologist, and one that should be heeded by present-day investigators, also pert•iins to odor and the emotional state (72): "If a certain degree of tumescence is required before a personal odor can exert an attractive influence, a powerful personal odor, strong enough to be perceived before any degree of tumescence is attained, will tend to cause repulsion." Despite the difficulties involved in gaining answers to the question of olfactory com- munication in primates, the quickening pace of research soon should enable us to form conclusions about learned odor associations. In the writer's opinion, the associative learning premise is potentially far more intriguing than the pheromonal model for human behavior. Although emotional responses of man are dominated by vision and hearing, olfaction certainly has not abdicated its function in shaping motivated be- havior. REFERENCES (1) S.N. Salthe, "Evolutionary Biology," Holt, Rinehart and Winston, Inc., New York, 1972. (2) J. B. S. Haldane, "The Causes of Evolution," Cornell University Press, Ithaca, NY, 1966. (3) K. B. Doving, Structure-activity relationships in chemoreception, Proceed/ngs of an ECRO Symposium, Benz, G. ed., Information Retrieval Ltd., London, 1976. (4) P. Karlson and A. Butenandt, Ann. Rev. Entomo/., 4, 39 (1959). (5) H. M. Bruce, J. Reprod. Ferti/., 1, 96 (1960). (6) P. Ropartz, C.R. Acad. So/., Paris 263, 2070 (1966). (7) S. Vander Lee and L. M. Boot, Acta physio/. pharmac. neer/., 4, 442 (1955). (8) W. K. Whirten, J. Endocrino/., 13,399 (1956). (9) M. S. Mayer and J. R. McLaughlin, "An Annotated Compendium of Insect Pheromones," Florida Agri- culture Experiment Stations Monograph Series #6, 1975. (10) D. D. Thiessen and M. Rice, Psycho/. Bull., 83,505 (1976). (11) R. P. Michael and E. B. Keverne, Nature (London), 218, 746 (1968). (12) R. P. Michael and E. B. Keverne, Nature (London), 225, 84 (1970). (13) R. P. Michael, E. B. Keverne, and R. W. Bonsall, Science, 172,964 (1971). (14) E. B. Keverne and R. P. Michael, J. Endocrino/., 51, 313 ( 1971).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)