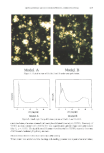
ENCAPSULATION INTO LIPOSOMES 347 3). Thus there was no binding of inulin to the surface of the liposomes that could yield an overestimation of EE. Furthermore, using this procedure, we were able to adequately separate free from encapsulated inulin (Figure 3). For these reasons, this method was selected for subsequent work. A disadvantage, however, is that liposome samples become contaminated with Ficoll © and are lost for use in further experiments. EVALUATION OF ENCAPSULATION EFFICIENCIES (EE) EEs obtained with various preparation methods and lipid concentrations are listed in Table I. RPE protocol 2 gave the highest EEs, with values ranging from 22-47%. These values are higher than EEs for inulin reported by other investigators (Table I) and compare well with those reported for other hydrophilic actives (Table II). Tables I and II also show wide variations in EE from one compound to another within a single preparation method. For example, liposomal gelonin prepared by RPE protocol 3 yielded EEs as high as 79% (Table II), whereas we found an EE of only 8% for liposomal inulin prepared by this method (Table I). Similar findings were noted earlier by Pidgeon et al. (24), who recommended that one always assay EE directly rather than relying on sucrose or carboxyfiuorescein data. Our results support this position. CONCLUSION The Ficoll © discontinuous density gradient method was found to be an excellent tool for separating free inulin from large, multilamellar liposome preparations. Using this method to determine encapsulation efficiencies (EE), we found that EE values approach- ing 40% were obtained by the reverse-phase evaporation (RPE) method, regardless of cholesterol level. The microstructure was shown to consist of large heterogeneous mul- tilamellar vesicles with diameters of 2-20 pro. At equal lipid concentration, much lower EE values (ca. 4%) were obtained for liposomes prepared by the film method. For inulin, comparison with other studies shows that EEs should be measured rather than inferred from systems having different compositions or preparation methods. ACKNOWLEDGMENTS We wish to thank Janet Burns for the cryo-TEM analysis, Brian Barford for assistance with the particle size measurements, and Dave Siegel for helpful discussions on liposome morphology. REFERENCES (1) K. Egbaria and N. Weiner, "Liposomes as a Topical Drug Delivery System Evaluated by In Vitro Diffusion Studies," in Cosmetic and Pharmaceutical Applications of Polymers, C. G. Gebelein, Ed. (Plenum Press, New York, 1991), pp. 215-224. (2) D. Yarosh, C. Bucana, C. Cox, L. Alas, J. Kibitel, and M. Kripke, Localization of liposomes containing a DNA repair enzyme in murine skin, J. Invest. Dermatol., 103, 461-468 (1994). (3) F. Bonina, L. Montenegro, C. La Rosa, F. Gasparri, and R. Leonardi, Comparison of different separative techniques in the quantitative determination of active compound enclosed in liposomal systems, Int. J. Cosmet. Sci., 16, 183-197 (1994).
348 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS (4) (5) (6) (7) (8) (9) (lO) (11) (12) (13) (14) (15) (16) (17) (18) (19) (20) (21) (22) (23) (24) (25) (26) (27) (28) A. Memoli, L. G. Palermiti, V. Travagli, and F. Alhaique, Liposomes in cosmetics: Which kind of phospholipid? Which loading method?,J. Soc. Cosmet. Chem., 44(2), 123-128 (1993). A. Memoli, L. G. Palermiti, V. Travagli, and F. Alhaique, Liposomes in cosmetics: II. Entrapment of a hydrophilic probe, J. Soc. Cosmet. Chem., 45(3), 167-172 (1994). A. Alam, S. R. K. Bhuri, A. K. Mavila, and V. Singh, Design of liposome to improve encapsulation efficiency of gelonin and its effect on immunoreactivity and ribosome inactivating property, Mol. Cell. Blochem., 112, 97-109 (1992). A. S. Verkman, R. Takla, B. Sefton, C. Basbaum, and J. H. Widdicombe, Quantitative fluorescence measurement of chloride transport mechanisms in phospholipid vesicles, Biochemistry, 28, 42404244 (1989). R. S. Kaplan and P. L. Pedersen, Isolation and reconstitution of the n-butylmalonate-sensitive dicar- boxylate transporter from rat liver mitochondria, J. Biol. Chem., 260, 10293-10298 (1985). J.A. Reynolds, Y. Nozaki, and C. Tanford, Gel-exclusion chromatography on S1000 Sephacryl: Application to phospholipid vesicles, Anal. Blochem., 130, 471-474 (1983). C. I. Price, J. W. Horton, and C. R. Baxter, Liposome encapsulation: A method for enhancing the effectiveness of local antibiotics, Surgery, 115,480-487 (1994). S. M. Goldin and S. W. Tong, Reconstitution of active transport catalyzed by the purified sodium and potassium ion-stimulated adenosine triphosphatase from canine renal medulla, J. Biol. Chem,, 249, 5907-5915 (1974). G. Blume, G. Cevc, M.D. J. A. Crommelin, I. A. J. M. Bakker-Woudenberg, C. Kluft, and G. Storm, Specific targeting with poly(ethylene glycol)-modified liposomes: Coupling of homing devices to the ends of the polymeric chains combines effective target binding with long circulation times, Blochim. Biophys. Acta, 1149, 180-184 (1993). D. W. Fry, J. C. White, and I. D. Goldman, Rapid separation of low molecular weight solutes from liposomes without dilution, Anal. Blochem., 90, 809-815 (1978). H. S. Penesky, A centrifuged-column procedure for the measurement of ligand binding by beef heart F1, Biochemistry, 18, 527-531 (1979). H. S. Penefsky, Reversible binding of Pi by beef heart mitochondrial adenosine triphosphatase,J. BioL Chem., 252, 2891-2899 (1977). A.D. Bangham, M. M. Standish, and J. C. Watkins, Diffusion of univalent ions across the lamellae of swollen phospholipids, J. Mol. Biol., 13, 238-252 (1965). D. Papahadjopoulos and J. C. Watkins, Phospholipid model membranes: Permeability properties of hydrated liquid crystals, Biochim. Biophys. Acta, 135,639-652 (1967). O. D. Gasko, A. F. Knowles, H. G. Shertzer, E.-M. Suolinna, and E. Racker, The use of ion-exchange resins for studying ion transport in biological systems, Biochemistry, 15, 57-65 (1976). R. Margalit, R. Alon, M. Linenberg, I. Rubin, T.J. Roseman, and R. W. Wood, Liposomal drug delivery: Thermodynamic and chemical kinetic considerations, J. Controlled Release, 17, 285-296 (1991). S. M. Gruner, R. P. Lenk, A. S. Janoff, and M. J. Ostro, Novel multilayered lipid vesicles: Comparison of physical characteristics of multilamellar liposomes and stable plurilamellar vesicles, Biochemistry, 24, 2833-2842 (1984). L. D. Mayer, M.J. Hope, P. R. Cullis, and A. S. Janoff, Solute distribution and trapping efficiencies observed in freeze-thawed multilamellar vesicles, Blochim. Biophys. Acta, 817, 193-196 (1985). R. Fraley, S. Subramani, P. Berg, and D. Papahadjopoulos, Introduction of liposome-encapsulated SV40 DNA into cells, J. Biol. Chem., 255, 10431-10435 (1980). T.D. Heath, B. A. Macher, and D. Papahadjopoulos, Covalent attachment of immunoglobulins to liposomes via glycosphingolipids, Blochim. Biophys. Acta, 640, 66-81 (1981). C. Pidgeon and S. McNeely, Multilayered vesicles prepared by reverse-phase evaporation: Liposome structure and optimum solute entrapment, Biochemistry, 26, 17-29 (1987). K. Egbaria and N. Weiner, Topical applications of liposomal preparations, Cosmet. Toiletr., 106, 79-93 (1991). K. Egbaria and N. Weiner, Liposomes as a topical drug delivery system, Adv. Drug. Deliv. Rev., 5, 287-300 (1990). D. Imbert, G. B. Kasting, and R. R. Wickett, Influence of liposomal encapsulation on the penetration of a large hydrophilic model compound through human skin in vitro, Pharm. Res., 12(9), S-281 (1995). A.D. Bangham, Physical structure and behaviour of lipids and lipid enzymes, Adv. Lipid. Res., 1, 65-104 (1963).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)