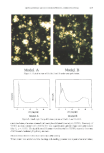
ENCAPSULATION INTO LIPOSOMES 343 .: , O:,lla O.1}am Figure 2. Cryo-TEM micrographs revealing several different vesicular morphologies observed in PC lipo- somes prepared by RPE protocol 2. Long, elongated structures are shear-induced artifacts of the observation method. The large honeycomb structure is part of the mounting apparatus. Arrows indicate inverted cubic phase. Magnification: 66,000x. density gradient separation method were tested for their ability to separate free inulin from liposomes. For each method, two test preparations were evaluated. A 2% w:v solution of inulin containing 14C-Carb-I was used to follow free inulin, and 14C-DPPC- labeled PC liposomes were used to evaluate liposome recovery. For the centrifuged-column method, the radiolabeled inulin solution was tested with Sephadex G-50 fine and Sephadex G-25 fine. At the first pass, 86% of total radiolabel was retained by the Sephadex G-50 column, whereas only 39% was retained by the Sephadex G-25. After two passes, 96% of radiolabel was retained by the Sephadex G-50 column and 65% was retained by the Sephadex G-25. Sephadex G-50 was therefore selected for the rest of the study and tested for its ability not to retain radioactivity associated with 14C-DPPC liposomes. Contrary to the results of other investigators (13,33,34), radiolabel recovery from the column eluant was extremely poor (less than 9%). Since most investigators reporting good results have worked with smaller lipo- somes (100-400 nm in diameter), we tested the hypothesis that the poor recovery was due to the large diameter of the liposomes studied. This was done qualitatively by sonicating liposome preparations, a common physical treatment to decrease liposome size. There was a positive correlation between sonication time and radiolabel recovery, indicating that the centrifuged-column method was not appropriate for large liposomes. Such limitations have been reported elsewhere (35). However, sonication or other physi- cal treatment aiming at decreasing liposome size was not a preferred alternative in our study since we felt it could yield leakage and significantly decrease EE. For this reason, the centrifuged-column method was not pursued further.
344 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS lOO 80- 60- 20- 0 • f 1 I 0 1 2 3 4 5 Cumulative Volume (m,) Top of centrifuge tube Bottom of centrifuge tube Figure 3. Determination of encapsulation efficiency by the discontinuous Ficoll © density gradient method. O, •4C-DPPC in PC liposomes prepared by RPE protocol 2 A, •4C-Carb-I in aqueous solution, [•, •4C-Carb-I + empty liposomes O, •4C-Carb-I partially entrapped in PC liposomes. Solid symbols represent fractions visually determined to contain liposomes. EE for the partially entrapped sample shown is 38%. In the dialysis method, ideally, liposomes are retained by the dialysis bag whereas free inulin diffuses out. In our study, there was no breakthrough of the liposomes after 24 hours ofdialysis. However, only 62-66% of inulin was recovered in the dialysate during this period. After 72 hours, recovery was 81-83%, rising to 83-85% after 95 hours. Such long dialysis times are inconvenient and also raise concerns about liposome leakage (31). Thus the high molecular weight and consequent slow diffusion of inulin limited the feasibility of the method. With the discontinuous FicolI © density gradient method, more than 95% of liposomes and less than 3% of free inulin were recovered in the top 1.5 ml of a 5-ml centrifuge tube. In most cases, however, the liposome fraction and less than 0.5 % of the free inulin were contained in a volume less than 400 pl. There was no significant difference in the distribution profiles of free inulin and free inulin mixed with empty liposomes (Figure
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)