SILICONE-BASED SUNSCREENS 379 DC-244 ........... 2.5% SV-5 .............. 2.5% Carbopol 1382 ...... 0.7% Nipaguard BPX ..... 0.5 % Triethanolamine ..... 08% DC-1401 .......... 3.0% Distilled water ..... c.s. 100 PREPARATION OF THE GEL EMULSION 1. Mix solar filters at 60 rpm with Finsolv SB (benzoic alcohol C12-C15), a solvent that ensures complete dissolution of Parsol 1789 in the silicone phase (RW20 stirrer, Janke and Kunkel GmbH, Staufen, Germany). 2. Add the cylomethicone mixture at 60 rpm to the combined filter solution. This yields an oily phase. 3. Disperse carbopol resin in the oily phase at 200 rpm. 4. Add distilled water very slowly with rapid shaking at 1700 rpm, under a vacuum if possible, until a milky, homogeneous dispersion is formed. 5. Neutralize the milky dispersion with triethanolamine. 6. Add dimethylconol slowly with rapid shaking at 1000 rpm. 7. Homogenize in a colloid mill (Lancor-Himmel, 2G.80 1.1H, Bilbao, Spain). RESULTS AND DISCUSSION SPECTROPHOTOMETRIC ANALYSIS The final formulation was analyzed, as was each of the solar filters separately. The results demonstrated the stability of the formulation under different experimental conditions. Figure 1 illustrates the absorption spectra of the UV-A and UV-B filters. Peak absorp- tions were found at 359 nm for Parsol 1789 and at 310 nm for Heliopan E-1000. Spectrophotometric analysis of the final cream (Figure 2) showed a major peak at 310 nm and a much weaker peak at 359 nm. Absorbance of longer wavelengths decreased steadily. These results were not surprising in view of the concentrations of the two solar filters in the final formulation: as much as five times more Heliopan E-1000 was present than Parsol 1789. The percentages of the two filters in the final formula were chosen in accordance with previously published recommendations and the limits set by current European law (10). The association of these two filters has the advantage of providing protection over a wide range of wavelengths and increasing the protective effect of our formulation. STABILITY ANALYSIS Because the preparations were thermodynamically unstable, we investigated viscosity and droplet size as well as organoleptic characteristics of the formulation. Tests were run
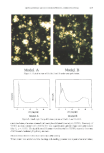
380 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS ABSORPTION ABSORPTION 1,6 - 1.2 - 0.9 0.6 - 0.3 - 359 nm. 1.5 310 nm. 200 240 260 320 360 400 WAVELENGLT•I (nm) i • PARSOL 1789 • HELlOPAN E-1000 • Figure 1. Absorption spectra of Parsol 1789 and Heliopan E-1000. 1.2 -0.9 -0.6 • 0 0.3 at different temperatures in accordance with the recommendations of Delonca and Roehl (11), and with the AFNOR T73 409, 1976 standard. Samples were kept at room temperature (25øC) for a period longer than six months, at 40øC for 12 weeks, and at 60øC for two weeks. Absorbance at the two wavelengths noted earlier was measured as a function of time. Figure 3 shows that there was very little difference in absorbance after exposure to each of the three temperature regimens. This result suggests that the solar filters in the preparation remained stable for these periods. To maintain the pH of skin, topical formulas should be slightly acidic. We therefore measured pH to check for variations with time. As Figure 4 shows, at room temperature the pH varied from a maximum of 7.3 in fresh preparations to a low of 6.94, remaining near 6.97 for most of the experimental period. At 40øC and 60øC, pH was approxi- mately 6.8 and never fell below 6.7. These figures also suggest that the preparations were stable. A series of rheological tests was done with samples at room temperature, 40øC and 60øC. The room temperature rheograms for the final formulation were similar throughout the 6-month study period. A hysteresis cycle was most prominent in samples tested at 60øC. Figure 5 shows rheograms for samples followed for six months at room temperature, for six weeks at 40øC, and for ten days at 60øC. Viscosity varied little at room temperature, but increased slightly with time at 40øC and 60øC. Rheograms showed that viscosity increased with time, reflecting a decrease in droplet size. These results were confirmed
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)