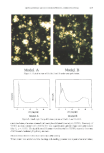
ENCAPSULATION INTO LIPOSOMES 341 et al. with a Ficoll © gradient (22). In our study, the protocol described by Fraley et al. (22) was adapted as follows: 0.6 ml of a 14C-labeled test preparation of known radio- chemical concentration was thoroughly mixed with 1.2 ml of Ficoll © 30%. One and one-half milliliters of the mixture was pipetted into a 5-ml polyallomer centrifuge tube (Beckman, Palo Alto, CA) and exactly weighed. Three milliliters of Ficoll © 10% were then carefully added on top of the first layer, followed by 0.5 ml of buffer. The tube was centrifuged at 100,000 x g for 30 min. Starting from the top of the centrifuge tube, 100- or 200-pl fractions of each layer were pipetted into scintillation vials and assayed for 14C by liquid scintillation counting. The presence of liposomes in these fractions was assessed visually. Fractions containing liposomes appeared milky, whereas non-liposome- containing fractions appeared clear. A solution containing 2% w:v inulin and 0.14 pCi/m114C-Carb-I in buffer was used to monitor the migration of inulin through Ficoll © upon ultracentrifugation. The migration of the liposomes was monitored separately using 14C-DPPC liposomes. Additionally, a solution containing 2% inulin, •4C-Carb-I, and empty PC liposomes (24 mg/ml) was also tested to ensure that there was no binding of inulin at the surface of the liposomes, which could result in an overestimation of EE. CALCULATION OF ENCAPSULATION EFFICIENCY (EE) EE is commonly defined as the number ratio of encapsulated molecules to total mol- ecules. To take into account variations that may simply arise from the amount of lipids used in liposomal preparations, some workers prefer to define EE as the amount of compound encapsulated per unit weight of lipids. In this study, we report both values. To calculate EE from Ficoll © separation data, the amounts of radioactivity (in CPM) recovered in each liposome fraction were pooled, and EE (in %) was determined using the following formula: EE (%) = 100' CPML/[AAs* m•] wherein CPM• is the total amount of radioactivity in liposome fractions (CPM), AA s is the average activity of the 30% Ficoll © layer before centrifugation (CPM/mg), and m• is the mass of the 30% Ficoll © layer in the centrifuge tube (mg). EE, expressed as mg inulin per mg lipid, was calculated as follows: EE (mg inulin/mg lipid) = lEE(%)* Cinulin]/CLipi D wherein Cinun n is the concentration of inulin (20 mg/ml) and CLiPi D is the concentration of phospholipid (variable, mg/ml). RESULTS AND DISCUSSION LIPOSOME CHARACTERIZATION Fresh PC and PC/Chol 9:1 mr liposomes prepared by RPE protocol 2 displayed similar size distributions, ranging from 2 to 20 lam (Figure 1). Analysis of a 21-day-old PC preparation showed a shift towards larger sizes (distribution between 2 and 100 lam). The shift for 15-day-old PC/Chol 9:1 mr liposomes was less significant (distribution between 3 and 30 lam). Such improvement in liposome long-term stability by addition of cholesterol was reported earlier (32). Vesicular structures displaying various mor-
342 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 2O 15- 10- _ 20 15- 10- _ i IIi I 0.1 PC liposomes 1.0 10.0 PClChol liposomes lOO - 80 - 60 - 40 - 20 - o I i i i i I 100.0 ii I i -- i i i i i i i 0.1 1.0 10.0 100.0 100 8O 6O 4O 2O Size (I.rm) Figure 1. Size distribution of fresh phospholipid liposome preparations prepared by RPE protocol 2• Top: PC liposomes bottom: PC/cholesterol 9:1 molar ratio. phologies were observed by cryo-TEM, including unilamellar and multilamellar lipo- somes, vesicles encapsulating smaller vesicles and tubular structures (Figure 2). SELECTION OF A SEPARATION METHOD The centrifuged-column separation method, dialysis, and the discontinuous Ficoll ©
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)