LIPO-AMINO ACID CHOLESTERYL DERIVATIVES 353 held in place by plastic tape (Tegaderm, 3M, Minnesota, USA). Prior to application of 1% SLS, the site was washed with a mild detergent (Minon Body Shampoo, Yamanouchi Pharmaceuticals Co., Ltd., Tokyo, Japan). Derreal scoring and application of samples. After 24-hour occlusive patch removal and rins- ing with water, TEWL and conductance were measured and each area was graded according to dermal scores. After induction of skin damage (after day 0), 2pl/cm 2 of cholesteryl derivatives were applied three times per day after the instrumental measure- ments and dermal scores gradings. The dermal scores were as follows: 0: No visible skin reaction 1: Barely perceptible erythema 2: Mild erythema 3: Well-defined erythema 4: Erythema and edema 5: Erythema and edema with vesiculation Experimental days were designated as follows: Day -1: dermal scores prior to induction of skin damage Day 0: dermal scores 30 minutes after patch removal Day 1: dermal scores 24 hours after induction of skin damage Day 2: dermal scores 48 hours after induction of skin damage Day 3: dermal scores 72 hours after induction of skin damage Conductance measurements and TEWL. The water-holding capacity of the stratum corneum was measured quantitatively using a skin surface hygrometer (11) (model Skicon 200, IBS Inc., Hamamatsu, Japan), which reads the conductance (pS) of the skin. Conduc- tance was measured five times at the same site, and the values were averaged to obtain individual values. TEWL was measured quantitatively with an evaporimeter (EP-1, ServoMed, Stockholm, Sweden). This instrument measures the vapor pressure gradient and is described in detail by Nilsson (13). The probe was held in place for each measurement until a stable TEWL value was established (approximately 30 seconds). TEWL values were then measured every 10 seconds for 50 seconds and were averaged to obtain individual values. APPLICATION OF CHOLESTERYL TEST MATERIALS ON DAMAGED SKIN (CHOLESTERYL DERIVATIVES AND ARTIFICIAL STRATUM CORNEUM LIPID MODELS)(11) 2pl/cm 2 of samples were applied to each patch site. Each sample was applied on the damaged skin daily for three days. Differences in measured values between samples were tested for significance using a Student t-test for paired samples. PREPARATION OF STRATUM CORNEUM LIPID The compositions of stratum corneum lipid model A (including ceramide) and model B (AGCE-301) are listed in Table I, based on Elias' analysis of the mammalian stratum comeurn (16). Models were prepared according to the method described by Friberg and
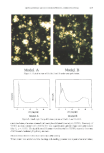
354 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Table I Composition of Model A and Model B Model A Model B wt.% Ceramide (type III) AGCE-301 18 Squalene Squalene 7 Triolein Triolein 25 Cholesterol sulfate Cholesterol sulfate 2 Cholesterol Cholesterol 14 Phosphatidyl ethanolamine Phosphatidyl ethanolamine 5 Pristane Pristane 4 Free acids* Free acids* 25 Total 100 * Constitution of free acids (wt%): oleic acid, 35 palmitic acid, 36 myristic acid, 15 stearic acid, 10 linoleic acid, 4. 4 t mean_+S.E. ' [ ' I ' I ' I ' -2 -1 0 1 2 3 4 days after application --' blank & AGCE-301 '" AGCE-202 []m Petrolatum - Cholesteryl Hydroxystearate Figure 1. Dermal scores following application of cholesteryl derivatives.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)