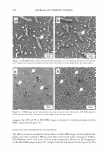
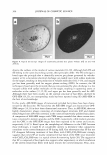
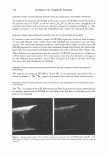
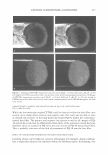
J. Cosmet. Sci., 56, 311-321 (September/October 2005) Effect of systemic hormonal cyclicity on skin NEELAM MUIZZUDDIN, KENNETH D. MARENUS, STEVEN F. SCHNITTGER, MICHAEL SULLIVAN, and DANIEL H. MAES, Estee Lauder Companies, 125 Pinelawn Road, Melville, NY 11747. Accepted for publication May 26, 2005. Synopsis Fluctuations in estrogen and progesterone during the menstrual cycle can cause changes in body systems other than the reproductive system. We conducted several studies to determine a possible correlation between phases of the menstrual cycle and specific skin properties. Healthy Caucasian women (ages 21-48), who had a typical 26-29 day menstrual cycle, participated in the studies. Measurements of skin barrier strength, dryness, response to lactic acid stinging, skin surface lipids, and microflora were obtained every week for two to three months. Ultraviolet B susceptibility in terms of minimal erythemal dose was also studied. The skin barrier was the weakest between days 22 and 26 of the cycle. Elevated neuronal response (lactic acid sting) was not observed to vary much with the cycle. Skin was driest between day 1 and day 6, while skin surface lipid secretion appeared to be highest on days 16-20 of the hormonal cycle. The highest microbial count was around days 16-22, and there was a high UV-B susceptibility between days 20 and 28 of the menstrual cycle. INTRODUCTION Menstrual cyclicity is a major biological process for women during their reproductive years and is associated with significant changes in hormonal status and behavior. An drogen excess from the ovaries or adrenal overproduction from the cutaneous metabolism of precursor hormones may be responsible for acne in some women (1-3). In addition to acne, other skin changes like skin thickness and echodensity (4), changes in transepi dermal water loss (TEWL) and blood flow (5-6), neuronal responses like pain (7), and irritant stimulus (8) have been implicated as consequences of hormonal changes. Clinical studies were designed to study changes in skin during the course of systemic fluctuations in estrogen and progesterone in a monthly menstrual cycle. In the first study, blood levels of progesterone and estradiol were measured once a week for a month. Additionally, barrier functions, neuronal responses, skin moisturization, and skin surface lipids and their correlation with hormonal changes in blood were studied. In Address all correspondence to N eelam Muizzuddin. 311
312 JOURNAL OF COSMETIC SCIENCE another study, the quantity of skin microflora was correlated with hormonal changes, and in a third study UV sensitivity was observed. MATERIALS AND METHODS SUBJECTS Females of ages 21-48, with no evidence of acute or chronic disease, including derma tological or ophthalmologic problems, were enrolled in the study. In order to qualify, the women had to be in a spontaneous ovulatory menstrual cycle. The subjects were not on any hormonal therapy or infertility treatment and had not taken any oral contraceptives for six months prior to the study. The facial skin had to be free of warts, nevi, moles, sunburn, suntan, scars, and active dermal lesions. On the day of the study, the subjects were instructed to refrain from using any lotions, creams, or other products on the face. Subjects were allowed to equilibrate for at least 30 minutes prior to testing in a controlled environment of 68 ° -70°F and 25% relative humidity. HORMONAL CYCLICITY Twenty-six female participants, ages 21-48, participated in the study. Blood was drawn from the subjects once a week at the same hour of the day by a trained technician following the standard operating procedures for phlebotomy at the clinical testing laboratory. The blood was centrifuged ( 100 rpm for 15 min) to pellet the blood cells and collect the serum. The serum was kept frozen until analyzed. Progesterone and estradiol were assayed from serum, employing a competitive immu noassay using direct chemiluminescence technology. Progesterone in the patient sample binds to an acridium ester-labeled mouse monoclonal anti-progesterone antibody. Un bound antibody binds to a progesterone derivative, covalently coupled to paramagnetic particles in the solid phase. Relative light units (RLU) detected by the system determine the quantity of progesterone. Estradiol in the patient sample competes with acridium ester-labeled estradiole in the reagent for a limited amount of rabbit anti-estradiol antibody in the antibody reagent. Rabbit anti-estradiole is captured by mouse anti-rabbit IgG, which is coupled to para magnetic particles in the solid phase. RLU detected by the system determine the quan tity of estradiol (9). EFFECTS OF SYSTEMIC HORMONAL FLUCTUATION ON SKIN The same participants as in the previous study used a simple TEA stearate-based lotion two weeks prior to commencement of the study and for the first month of the study. The subjects applied the assigned test materials on the full face twice a day and were instructed not to change their daily cleansing regimen and makeup for the course of the study. The subjects reported to the clinical testing facility once a week at the same hour of the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)