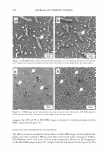
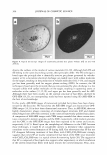
2005 ANNUAL SCIENTIFIC SEMINAR 371 Using D&C Black 2 The approved uses for carbon black for eyeliner, brush-on brow makeup, eye shadow, mascara, lipstick, blushers and rouge, makeup and foundation and nail enamel. Of course, due to the jet-black color of carbon black, the most popular uses will probably be in the area of mascaras and eyeliners, where the intensity of color obtainable will make it most desirable. When carbon black was originally delisted, it was replaced by iron oxide black, which, by comparison, is less intense and less jet-black, often appearing gray or brown. Table 2 compares the properties of carbon black and black iron oxide: I Property I Carbon Black I Black Iron Oxide Formula C Fe· FeO2 ~20-30 nm ~ 300 nm * The Chemistry and Manufacture of Cosmetics, M deNavarre, ed, Wheaton, Ill: Allured publishing ( 1971 ). This value, from 30 years ago was specifically for carbon black, and may not apply in the case ofD&C Black #2. Table 2. Comparison of Carbon Black and Black Iron Oxide The stability of carbon black is a great advantage over iron oxide. Often times, the heat generated by processing alone, especially in powder applications, using micronizers, would be enough to cause oxidation to the red form. Aside from the obvious aesthetic problem of shade, enough heat could be generated to cause cardboard drums, often used for pack-out, to smolder or bum. Due to its reactivity to heat, iron oxide black is classified as a hazardous material, and demands special shipping considerations. And, of course, the tinting strength of carbon black, at least 2 - 3 times that of typical black iron oxides is its biggest advantage. Of course, there are disadvantages to the use of carbon black as well. Due to the fineness of particle size, it is very "fluffy" and can easily become suspended in the environment of the workplace if not handled with extreme caution. The high surface area makes it very adsorbent in most liquid applications, resulting in relatively high viscosity. In most carriers, it is difficult to obtain dispersions of more than 20-25%. Fortunately, the increased intensity allows for lower use levels, minimizing the viscosity increase experienced in applications like mascaras and eyeliners. Conclusion If interest in carbon black as a colorant can be maintained due to its advantages, it is possible that new wetting agents and/or surface treatments may, in time, be developed which reduce the difficulty of working with it. In any event, it is nice to see that the relisting of carbon black, even with the imposed restrictions, indicates that the delisting of a colorant may not mean that it is gone forever.
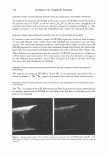
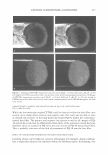
372 JOURNAL OF COSMETIC SCIENCE PERMANENT WAVING AND DEPILATION R. Randall Wickett, Ph.D. University of Cincinnati, College of Pharmacy, 3223 Eden Ave, Cincinnati, OH 45267 Introduction Permanent waves and depilatories reply on the breaking of hair keratin disulfide bonds by mercaptans. Salts of thioglycolic acid (TGA) are most often used to achieve this purpose. From the standpoint of the chemistry involved the main differences between depilatories and perms are pH and counter ion and the fact that perms require reforming the s�s bond through an oxidative step(1 ). Perms using thioglycolate are typically formulated between pH 9 and 9.6. The so-called acid perms that utilize glycerol monothioglycolate (GMT) are typically adjusted to pH 6.8-7.0(2). The ammonium salt is most often used with TGA to provide swelling of the hair. Hydrogen peroxide is most commonly used for neutralization. Depilatories are typically formulated at pH 12-12.5 and utilize calcium or sodium salts or a mixture of the two(3). Chemistry and Physics of Hair Reduction for waving and depilation Wolfram and Underwood described the reactions between reducing agents and keratin disulfide bonds in detaiI(4). With TG the active species is the thiolate ion, RS-(1 4). With monothiol reducing agents the reaction proceeds in two steps converting two moles of the reducing agent to disulfide for each reduced S-S bond. 1. Ker-5-5-Ker + R-5- � Ker-5-5-R + Keratin Mercaptan Mixed disulfide 2. Ker-5-5-R + R-5- � R-5-5-R + Mixed disulfide Mercaptan disulfide Ker-5- Reduced Keratin Ker-s· Reduced keratin Reduction of the S-S bonds leads to structural changes that are keys to the action of perms and depilatories. Depilatory action is achieved when the hair is weakened to the point that it may be rubbed off of the skin. With perms the key is to mobilize the structure while the hair is under stress so that alterations in the polymeric structure can be locked in by the neutralizer treatment to produce permanent set. This is achieved by breaking a fraction of the disulfide bonds and assisted by the presence of free SH groups in the hair which can participate in the sulfide disulfide interchange reaction(1 5). It can be argued that the key bonds that must be broken for either depilation or permanent waving are those that support tensile stress when the hair is under load{6-8). This makes chemical stress relaxation methods very useful for studying reduction kinetics related to either process(6 8-12). To use these methods a hair is placed under tension in buffer and stress relaxed until it supports a constant level of force. The buffer is then switched for the reducing agent and the decay of the force with time is followed.· This method, also called Single Fiber Tensile Kinetics (SFTK) has been used to study the effects of pH, temperature and hair type on the reaction kinetics(6) and to study the effect of reducing agent structure. (6 13) on reduction rates. I have postulated that the reaction kinetics follow at least two different mechanisms depending on pH and reducing agent used(7 11 ). For example with TGA reaction kinetics apparently follow a pseudo first order mechanism at low pH while at higher pH reaction proceeds by a sharp front or "moving boundary". The SFTK method has been applied to the study of depilatories as well as perms. For depilatories the time required for the tensile force to decay to 5% of its original value was found to be a convenient indicator of depilatory activity.(11) that correlated well with depilation times measured in vivo. Effects of pH, counter ion and pretreatment were quantified. Beidman(14) used a thermomechanical analyzer to determine the time required for the stretching of a hair bundle in depilatory solution to begin and also reported good correlation with in vivo depilatory efficacy.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)