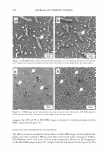
2005 ANNUAL SCIENTIFIC SEMINAR 373 We have also compared SFTK data to data obtained using amino acid analysis for TGA and cysteamine(9) and concluded that while SFTK kinetics appear somewhat faster than kinetics determined by amino acid analysis the rank order of the reaction is consistent between the two methods. Wortmann and Souren(12) used an innovative combination of chemical stress relaxation and intermittent strain pulses to try to correlate changes in tensile properties with the level of permanent set developed. The reaction was followed during both the reduction and oxidation steps. While the relaxed tensile force will not increase during oxidation the increase in modulus measured from the small strain pulses will be dependant on the number of S-S bonds reformed. The set recovery of hair subjected to the same treatment protocol was measured by treating the hair on a small cylinder of known diameter De, then measuring the diameter, D to which the hair loop opened in water. They reported that recovery, R, the opposite of set, is directly proportion to the ratio of the fraction tensile strength lost during the reduction step to the fraction of tensile strength regained during the oxidation step. The efficacy of perms may also be evaluated using the pegboard (15) or the test tube curl method(15-17). While direct kinetic data can't be obtained from these methods they are very useful for laboratory evaluation of permanetlt waves. Unfortunately the waving process does do some damage to hair. A small but significant reduction in tensile strength occurs(2 18 19). Recent results by Nishikawa et al indicate that the content of a-helical protein is reduced from 23.7% to 20.4% and that the resultant conformational change is to random coil. Reference List 1. Albrecht, L. and Wolfram L.J. (1982) J Soc Cosmet Chem 33, 363-366 2. Wickett, R. R. and Savaides, A. (2001) Permanent Waving of Hair. In Schlossman, M., editor. Chemistry and Manufacture of Cosmetics, Volume II, Formulating, Allured Publishing, Carol Stream II. 3. Shea, F. T. (2001) Depilatories and Epilatories. In Schlossman, M. , editor. Chemistry and Manufacture of Cosmetics, Volume II, Formulating , Allured Publishing, Carol Stream, II 4. Wolfram L.J. and Underwood, D. L. (1966) Textile Res J 36, 947-953 5. Feughelman, M. (1990) J Soc Cosmet Chem 41, 209-212 6. Wickett, R.R. (1983) J Soc Cosmet Chem 34, 301-316 7. Wickett, R.R. (1991) Cosmetics and Toiletries 106, 37-47 8. Wortmann, F. J. and Souren, I. (1986) J Soc Cosmet Chem 37, 461-473 9. Manuszak, M.A., Berish, E.T., and Wickett, R. R. (1996) J Soc Cosmet Chem 47, 213-228 10. Manuszak, M.A., Berish, E. T., and Wickett, R.R. (1996) J Soc Cosmet Chem 47, 49-58 11. Wickett, R. R. and Mermelstein, R. (1986) J Soc Cosmet Chem 37, 461-4 73 12. Wortmann, F. J. and Kure, N. (1990) J Soc Cosmet Chem 41, 123-139 13. Wickett, R.R.and Barman, B. G. (1985) J Soc Cosmet Chem 36, 75-86 14. Beideman, F. E. (1987) J Soc Cosmet Chem 38, 287-294 15. Kirby, D. H. (1956) Proc Sci Sect Toilet Goods Assoc 26, 12-15 16. Haefele, J. W. (1955) Hair Waving Lotion .. United States Patent 2,719,814) 17. Marti, M. E. (1990) Cosmetics and Toiletries 105, 113-120 18. Robbins C.R. (1994) The physical properites and cosmetic behavior of hair. In Robbins C.R., editor. Chemical and Physical Behavior of Human Hair, Springer-Verlag, New York 19. Wickett, R. R. (1995) Measuring the Mechanical Strength of Hair. In J.Serup and B.E.Jemec, editors. Handbook of Non-Invasive Methods and the Skin, CRC Press, Boca Raton
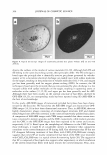
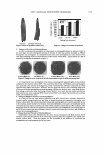
374 JOURNAL OF COSMETIC SCIENCE A NOVEL PHOSPHATE ESTER FOR HAIR-COLORING ENHANCEMENT Timothy Gao, Ph.D., Abel Pereira, Yoon-Hee Choi, Jung-Mei Tien and Robert Lanese Croda, Inc., 300-A Columbus Circle, Edison, NJ 08837 Introduction Bleaching and coloring of hair has become increasingly popular over the past years. Color is produced inside the hair fiber by hydrogen peroxide-induced oxidation and coupling reactions of aromatic amines and phenols. Since hydrogen peroxide is used, it will concurrently bleach the hair's melanin, and shades lighter than the natural hair color can be obtained. Oxidative dyeing is usually carried out under alkaline conditions preferably at pH 9 to I 0. At the time of dyeing, the alkaline solution containing the dye components is mixed with the peroxide developer, which is stabilized for storage at a pH of 3 to 5. The resulting mixture is then applied to hair for 20 to 40 minutes before being rinsed off with water. Surfactants play important roles in the efficacy of hair color products: Emulsification - to form stable and fine O/W emulsions for better solubility and distribution of dye intermediates and coupling agents Wetting - to accelerate the diffusion of dye intermediates and coupling agents into hair cortex Cleaning and Spreading - to ensure uniform dye coverage on the air surface and help removing dye residue on the hair surface, which contributes to a poor feel, dull appearance, and difficult combing. There have been a lot of studies on the factors that affect the bleaching and coloring process. These factors include dye solubility and distribution, coupling rates between dye and coupling agent, diffusion rates of dye intermediates and coupling agents into the hair cortex, hair swelling rate, effect of different alkali on dyeing process, and the efficiency of dye use. Phosphate ester compounds have long been used in hair and skin care formulations including hair relaxers, hair perms, sunscreens, and color bases as excellent emulsifiers and viscosity thickening agents. In this article we report our recent findings on how phosphate esters affect oxidative dye coloring performance. An excellent Hair Color Enhancer (HCE) - Oleth-5 Phosphate and Dioleyl Phosphate has been found. Experimental Materials I. Hair Dye bases were obtained from the Applications Lab at Innovation Center of Croda Inc Color Base A - A regular oxidative auburn color base without phosphates Color Base B - A modified Color Base A with addition of 4% HCE 2. Hydrogen Peroxide Hair Color Developer was prepared by Application Lab of Croda Inc 3. Bleached Hair was purchased from International Hair Importers and Products, Inc, New York Measurements 1. Hair color indexes {L, a•, b*) were determined using a LabScan XE Spectrocolorimeter equipped with a special sample holder for hair tress. 2. Dynamic advancing contact angle at the interface of hair fiber/deionized water was determined using a Cahn DCA-315 Dynamic Contact Angle Analyzer at constant temperature of 25°C. 3. Emulsion structure was inspected under a Nikon Optiphot -Pol Microscope, and the image was saved and analyzed by image analysis software. 4. Viscosity profile of emulsion samples was measured using a Brookfield DY-III Rheometer at constant temperature of 25°C Results and Discussion 1. Change in Hair Color with Time The photos of dyed hair samples are shown in Figure 1. The determined changes in hair red color index, a•, of bleached hair before and after dyeing with the two different dye bases are presented in Figure 2. It can be easily seen by visual inspection that the hair dyed with the color base containing HCE showed richer red color than the hair dyed with a regular base. The determined changes in the red color index of Lia*, in yellowing index of Lib*, and in total color difference of �E of these hair tresses clearly indicated that the development rate of red color on the hair dyed with the color base containing additional HCE was faster than that on the hair dyed with the base without HCE. This experimental result indicates that addition of HCE into regular oxidative auburn color base not only produced richer final shade, but also accelerated the whole coloring process and demonstrated faster coloring rate compared to the hair dyed with the corresponding regular dye without HCE.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)